Back
BackChapter 5: Molecules and Compounds – Structured Study Notes
Study Guide - Smart Notes

Molecules and Compounds
Sugar and Salt: Properties of Compounds vs. Elements
Compounds are substances formed from two or more elements chemically combined in fixed ratios. Their properties are often very different from the properties of the individual elements that compose them. For example, sucrose (table sugar) is a compound made of carbon, hydrogen, and oxygen, while sodium chloride (table salt) is formed from sodium and chlorine.
Compound: A substance composed of two or more elements in fixed, definite proportions.
Example: Sucrose (C12H22O11) and sodium chloride (NaCl).
Contrast: The properties of compounds differ greatly from their constituent elements (e.g., sodium is a reactive metal, chlorine is a poisonous gas, but NaCl is safe table salt).





Compounds Display Constant Composition
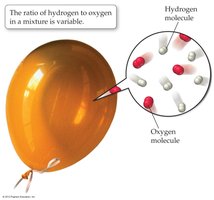
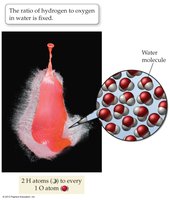
Most substances encountered in daily life are compounds, not elements. Compounds are characterized by the law of constant composition, which states that all samples of a given compound have the same proportions of their constituent elements.
Law of Constant Composition: All samples of a compound have the same proportions of elements.
Mixture vs. Compound: Mixtures can have variable proportions; compounds have fixed ratios.
Example: Water always has a hydrogen-to-oxygen ratio of 2:1.
Constant Composition: Mass Ratio
When compounds are decomposed, the mass ratios of their constituent elements are constant. For example, decomposing water yields a fixed mass ratio of oxygen to hydrogen.
Water: or
Ammonia: or
Atoms combine in whole-number ratios, but mass ratios are not necessarily whole numbers.
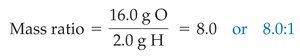
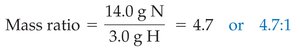
Chemical Formulas: Representing Compounds
How to Represent Compounds
Chemical formulas indicate the elements present in a compound and the relative number of atoms of each. Subscripts show the number of atoms; a subscript of 1 is omitted.
Examples: H2O (water), NaCl (table salt), CO2 (carbon dioxide), C12H22O11 (sucrose).
Subscripts are part of the compound’s definition; changing them creates a different compound (e.g., CO vs. CO2).
Order of Listing Elements in Chemical Formulas
Formulas list the most metallic element first. For compounds without metals, the more metal-like element is listed first. Among nonmetals, those to the left and bottom of the periodic table are more metal-like.
Example: NaCl, not ClNa; NO2, not O2N.

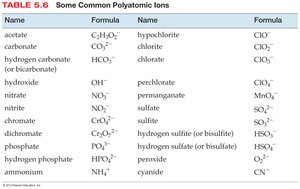
Polyatomic Ions in Chemical Formulas
Some chemical formulas contain groups of atoms that act as a unit, called polyatomic ions. Parentheses and subscripts indicate the number of each group.
Example: Mg(NO3)2 contains one magnesium atom and two nitrate groups.
To find the total number of each atom, multiply the subscript outside the parentheses by the subscript inside.
Types of Chemical Formulas
There are three main types of chemical formulas:
Empirical Formula: Shows the simplest ratio of elements (e.g., HO for hydrogen peroxide).
Molecular Formula: Shows the actual number of atoms (e.g., H2O2).
Structural Formula: Shows how atoms are connected.

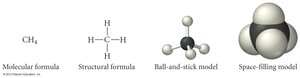
Representations of Molecules
Molecular models help visualize the structure and geometry of molecules. Ball-and-stick models show connections, while space-filling models show the actual space occupied by atoms.
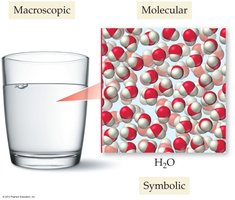
A Molecular View of Elements and Compounds
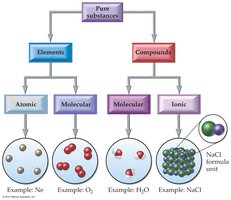
Classification of Pure Substances
Pure substances can be elements or compounds. Elements may be atomic or molecular, while compounds may be molecular or ionic.
Atomic Elements: Exist as single atoms (e.g., Ne, Hg).
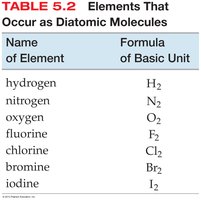
Molecular Elements: Exist as diatomic molecules (e.g., O2, Cl2).
Molecular Compounds: Composed of molecules (e.g., CO2).
Ionic Compounds: Composed of cations and anions (e.g., NaCl).
Atomic and Molecular Elements
Atomic elements exist as individual atoms, while molecular elements exist as diatomic molecules.
Example: Mercury (Hg) is atomic; chlorine (Cl2) is molecular.

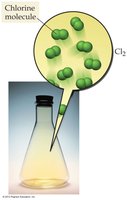
Molecular and Ionic Compounds
Molecular compounds are formed from nonmetals and consist of molecules. Ionic compounds are formed from metals and nonmetals, consisting of cations and anions arranged in a lattice.
Example: Dry ice (CO2) is molecular; table salt (NaCl) is ionic.
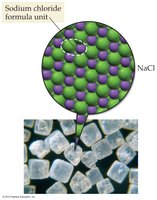
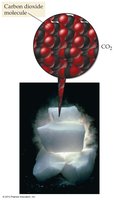
Ionic Compounds
Ionic compounds are formed when metals lose electrons and nonmetals gain electrons, resulting in positive and negative ions held together by electrostatic attraction. The basic unit is the formula unit, not a molecule.
Writing Formulas for Ionic Compounds
Rules for Writing Ionic Compound Formulas
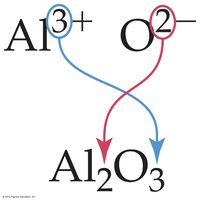
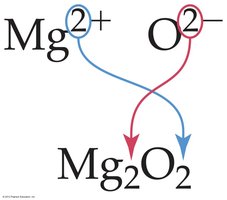
Ionic compounds must be electrically neutral. The sum of the charges of the cations and anions must equal zero.
Write the symbol and charge for each ion.
Use the magnitude of each ion’s charge as the subscript for the other ion.
Reduce subscripts to the smallest whole-number ratio.
Nomenclature: Naming Compounds
Naming Ionic Compounds
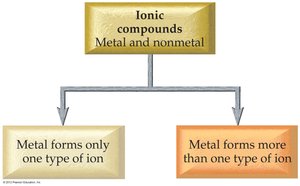
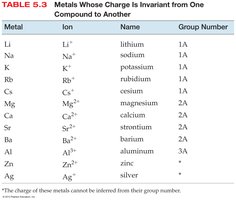
Ionic compounds are named based on the type of metal present. Type I metals have invariant charges; Type II metals can form more than one type of ion.
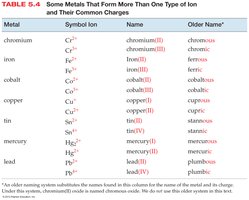
Naming Binary Ionic Compounds (Type I)
For compounds with metals that form only one type of ion, the name consists of the cation name followed by the base name of the anion with the ending -ide.
Example: NaCl is sodium chloride.

Naming Binary Ionic Compounds (Type II)
For compounds with metals that form more than one type of ion, the charge of the metal is specified with a Roman numeral in parentheses.
Example: FeCl3 is iron(III) chloride.

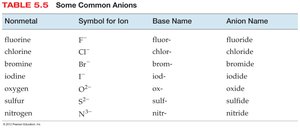
Naming Ionic Compounds with Polyatomic Ions
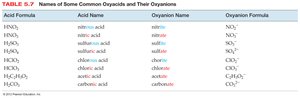
When a polyatomic ion is present, use its name in the compound name. Many polyatomic ions are oxyanions, named systematically based on the number of oxygen atoms.
-ate: More oxygen atoms (e.g., sulfate SO42−)
-ite: Fewer oxygen atoms (e.g., sulfite SO32−)
Prefixes hypo- and per- are used for series with more than two ions.
Everyday Chemistry: Polyatomic Ions
Polyatomic ions are found in many household products, such as bleach (sodium hypochlorite), baking soda (sodium bicarbonate), antacids (calcium carbonate), and food preservatives (sodium nitrite).

Naming Molecular Compounds
Rules for Naming Molecular Compounds
Molecular compounds are formed from two or more nonmetals. Their names use prefixes to indicate the number of atoms of each element.
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-
If only one atom of the first element, the prefix mono- is omitted.
Example: CO2 is carbon dioxide; N2O is dinitrogen monoxide.

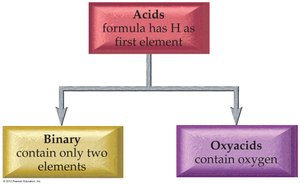
Naming Acids
Classification and Naming of Acids
Acids are molecular compounds that produce ions in water. They are categorized as binary acids (hydrogen and a nonmetal) or oxyacids (hydrogen, a nonmetal, and oxygen).
Naming Binary Acids
Binary acids are named with the prefix hydro-, the base name of the nonmetal, and the suffix -ic, followed by the word acid.
Example: HCl(aq) is hydrochloric acid.

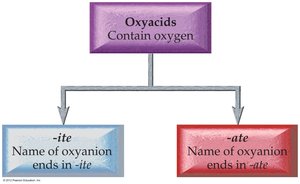
Naming Oxyacids
Oxyacids contain oxyanions. If the oxyanion ends in -ite, the acid name ends in -ous; if it ends in -ate, the acid name ends in -ic.
Example: HNO2 (nitrite) is nitrous acid; HNO3 (nitrate) is nitric acid.

Chemistry in the Environment: Acid Rain
Acid rain is caused by rainwater mixing with air pollutants such as NO, NO2, and SO2, which form acids like HNO3 and H2SO4. Acid rain harms aquatic life, weakens trees, and damages buildings.
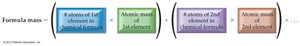
Formula Mass: The Mass of a Molecule or Formula Unit
Calculating Formula Mass
The formula mass of a compound is the sum of the atomic masses of all atoms in its chemical formula.
Formula:
Example: Calculate the formula mass of Al2(SO4)3.