 Back
BackChapter 5: Molecules and Compounds – Structured Study Notes
Study Guide - Smart Notes

Molecules and Compounds - CH.5
Introduction to Compounds
Compounds are substances composed of two or more elements chemically combined in fixed proportions. The properties of compounds are generally very different from the properties of the elements that compose them. For example, table sugar (sucrose) and table salt (sodium chloride) have properties distinct from their constituent elements.
Compound: A substance formed when two or more elements are chemically bonded in fixed ratios.
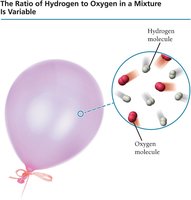
Mixture: A combination of substances in variable proportions, not chemically bonded.
Example: Sucrose contains carbon, hydrogen, and oxygen, but its properties differ from graphite (carbon), hydrogen gas, and oxygen gas.
Constant Composition and the Law of Constant Composition
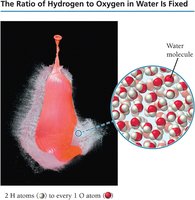
Compounds display constant composition, meaning the elements combine in fixed, definite proportions. This is formalized in the law of constant composition, which states that all samples of a given compound have the same proportions of their constituent elements.
Law of Constant Composition: All samples of a compound have the same proportions of elements by mass.
Example: Water always decomposes into 16.0 g of oxygen and 2.0 g of hydrogen for every 18.0 g sample.
Mixtures: The ratio of elements can vary, as shown in a balloon filled with hydrogen and oxygen.
Chemical Formulas
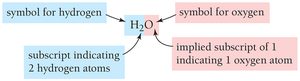
Chemical formulas represent compounds by indicating the elements present and the relative number of atoms of each. Subscripts show the number of atoms; a subscript of 1 is omitted by convention.
Empirical Formula: Simplest whole-number ratio of atoms.
Molecular Formula: Actual number of atoms in a molecule.
Structural Formula: Shows how atoms are connected.
Example: Water is represented as H2O.
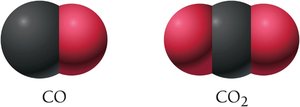
Changing a subscript in a chemical formula results in a different compound. For example, CO (carbon monoxide) and CO2 (carbon dioxide) are distinct compounds.
Order of Elements in Chemical Formulas
The order of elements in a chemical formula is determined by their metallic character. The most metallic element is listed first. Among nonmetals, those to the left or lower in the periodic table are listed first.
Example: NaCl (sodium chloride), not ClNa.
Polyatomic ions: Groups of atoms that act as a unit and may have a charge.
Polyatomic Ions
Polyatomic ions are groups of atoms with an overall charge that act as a single unit in compounds. When more than one polyatomic ion is present, parentheses and subscripts are used to indicate quantity.
Example: Mg(NO3)2 contains one magnesium ion and two nitrate ions.
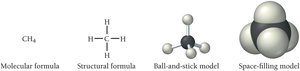
Types of Chemical Formulas and Models
Chemical formulas can be empirical, molecular, or structural. Models such as ball-and-stick and space-filling illustrate the 3D structure of molecules.
Empirical formula: H2O for water.
Molecular formula: C12H22O11 for sucrose.
Structural formula: Shows connectivity.
Ball-and-stick model: Shows geometry.
Space-filling model: Shows how atoms fill space.
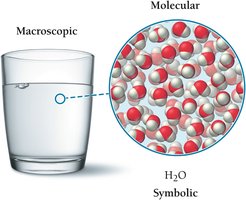
Connecting Macroscopic and Molecular Worlds
Structural formulas and models help connect the macroscopic world (what we see) to the atomic and molecular world (the particles that compose matter).
Classification of Elements and Compounds
Elements: Atomic vs. Molecular
Elements can exist as atomic or molecular forms. Atomic elements exist as single atoms, while molecular elements exist as molecules, often diatomic (two atoms bonded together).
Atomic elements: Mercury (Hg) exists as single atoms.
Molecular elements: Chlorine (Cl2) exists as diatomic molecules.

Compounds: Molecular vs. Ionic
Compounds are classified as molecular or ionic. Molecular compounds are formed from nonmetals and consist of molecules. Ionic compounds are formed from metals and nonmetals, consisting of cations and anions in a 3D array.
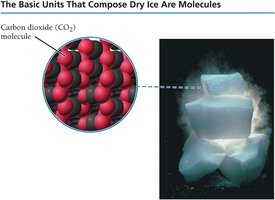
Molecular compounds: CO2 (dry ice) is composed of molecules.
Ionic compounds: NaCl (table salt) is composed of sodium and chloride ions.
Writing and Naming Chemical Formulas
Writing Formulas for Ionic Compounds
Ionic compounds are formed when metals bond to nonmetals. The sum of the charges of cations and anions must be zero. The formula is determined by balancing the charges.
Example: Na+ and Cl- combine to form NaCl.
Example: Mg2+ and Cl- combine to form MgCl2.
Polyatomic Ions in Formulas
When writing formulas with polyatomic ions, use parentheses if more than one polyatomic ion is present. The subscript outside the parentheses indicates the number of polyatomic ions.
Example: Ca2+ and NO3- combine to form Ca(NO3)2.
Naming Ionic Compounds
Ionic compounds are named based on the type of metal present. Type I compounds contain metals with constant charges (main group elements), while Type II compounds contain metals with variable charges (transition metals).
Type I: Name the cation (element name) and the anion (base name + -ide).
Type II: Name the cation (element name + Roman numeral for charge) and the anion (base name + -ide).
Example: NaCl is sodium chloride; FeCl3 is iron(III) chloride.
Naming Ionic Compounds with Polyatomic Ions
Use the name of the polyatomic ion whenever it occurs in the compound. If both cation and anion are polyatomic, use both names.
Example: KNO3 is potassium nitrate; NH4NO3 is ammonium nitrate.
Naming Oxyanions
Oxyanions are anions containing oxygen. The naming depends on the number of oxygen atoms:
-ate: More oxygen atoms (e.g., nitrate).
-ite: Fewer oxygen atoms (e.g., nitrite).
Prefixes: hypo- (less than), per- (more than).
Polyatomic Ions in Everyday Chemistry
Polyatomic ions are found in many household products. Sodium hypochlorite is the active ingredient in bleach, sodium bicarbonate is found in baking soda, and calcium carbonate is used in antacids.


Naming Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of atoms. The first element is the more metal-like one, and the second element's name ends with -ide.
Prefixes: mono-, di-, tri-, tetra-, penta-, etc.
Example: CO2 is carbon dioxide; N2O is dinitrogen monoxide.
Exception: The prefix mono- is omitted for the first element.
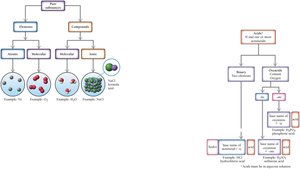
Naming Acids
Acids are molecular compounds that form ions in water. They are classified as binary acids (hydrogen and a nonmetal) or oxyacids (hydrogen, a nonmetal, and oxygen).
Binary acids: Named as hydro- + base name + -ic acid (e.g., hydrochloric acid).
Oxyacids: Named based on the oxyanion present. If the oxyanion ends in -ate, the acid name ends in -ic; if it ends in -ite, the acid name ends in -ous.
Nomenclature Flowchart
Nomenclature flowcharts help determine the correct naming system for compounds, acids, and ions.
Formula Mass
Calculating Formula Mass
The formula mass of a compound is the sum of the atomic masses of all atoms in its chemical formula. This is important for quantitative chemical calculations.
Formula:
Example: MgCl2 formula mass is calculated by adding the atomic masses of Mg and two Cl atoms.
