 Back
BackChapter 6: Chemical Composition – Study Guide
Study Guide - Smart Notes

Chemical Composition
Introduction to Chemical Composition
Chemical composition refers to the types and amounts of elements that make up a compound. Understanding chemical composition is essential for quantifying substances, predicting reactions, and analyzing materials in chemistry.
The Mole Concept
Definition and Importance
The mole is a fundamental unit in chemistry used to count atoms, molecules, or ions. One mole contains 6.022 × 1023 entities, known as Avogadro’s number. This allows chemists to relate microscopic particles to macroscopic quantities.
1 mole = 6.022 × 1023 units (atoms, molecules, ions, etc.)
The mole bridges the gap between atomic scale and laboratory scale measurements.
Example: 1 mole of copper atoms is approximately the number of atoms in 22 pennies.


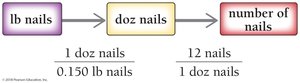
Counting by Weighing: Analogy
Counting atoms is analogous to counting objects by weight, such as nails sold by the pound. Conversion factors are used to relate mass to number of items.
For nails: Use weight per dozen and number per dozen as conversion factors.
For atoms: Use molar mass and Avogadro’s number as conversion factors.
Atomic Mass and Molar Mass
Definitions
The atomic mass unit (amu) is defined as one-twelfth the mass of a carbon-12 atom. The molar mass of an element is the mass of 1 mole of its atoms, expressed in grams per mole (g/mol).
Atomic mass (amu) is used for individual atoms.
Molar mass (g/mol) is used for bulk quantities.
Example: Copper has an atomic mass of 63.55 amu and a molar mass of 63.55 g/mol.

Conversions: Moles, Grams, and Atoms
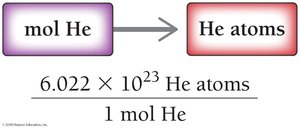
Converting Moles to Number of Atoms
To convert moles to number of atoms, multiply by Avogadro’s number:
Example: 3.5 mol He × 6.022 × 1023 = 2.1 × 1024 He atoms
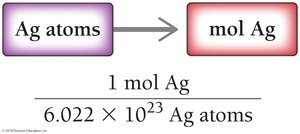
Converting Number of Atoms to Moles
To convert number of atoms to moles, divide by Avogadro’s number:
Example: Ag atoms ÷ = mol Ag
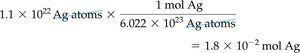
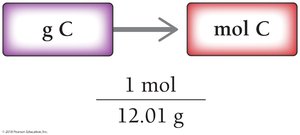
Converting Grams to Moles
To convert grams to moles, use the molar mass as a conversion factor:
Example: 0.58 g C ÷ 12.01 g/mol = 0.048 mol C
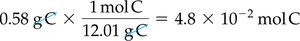
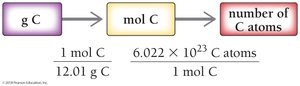
Converting Grams to Moles to Atoms
To find the number of atoms in a given mass:
Step 1: Convert grams to moles using molar mass.
Step 2: Convert moles to atoms using Avogadro’s number.
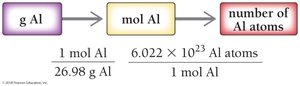
Converting Grams to Atoms: Example with Aluminum
For a 16.2 g aluminum can:
Step 1:
Step 2:
Result: Al atoms
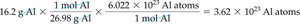
Molar Mass of Compounds
Definition and Calculation
The molar mass of a compound is the mass of 1 mole of its molecules or formula units. It is calculated by summing the atomic masses of all atoms in the chemical formula.
For water:
For NO2:
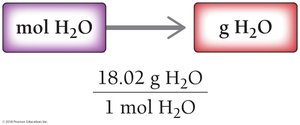
Converting Between Grams and Moles of a Compound
To convert moles to grams, multiply by the molar mass:
Example: 1.75 mol H2O × 18.02 g/mol = 31.5 g H2O
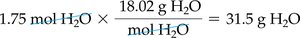
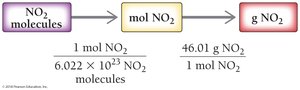
Converting Number of Molecules to Mass
To convert molecules to mass:
Step 1: Convert molecules to moles using Avogadro’s number.
Step 2: Convert moles to grams using molar mass.
Example: 4.78 × 1024 NO2 molecules = 365 g NO2
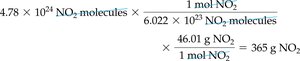
Chemical Formulas as Conversion Factors
Using Chemical Formulas
Chemical formulas indicate the ratio of elements in a compound. These ratios can be used as conversion factors between moles of compound and moles of constituent elements.
Example: CO2 has 2 O atoms per 1 CO2 molecule.
Conversion factor: 2 mol O : 1 mol CO2
Converting Between Moles of Compound and Moles of Element
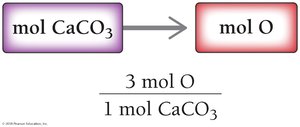
To find moles of an element in a compound:
Use the ratio from the chemical formula.
Example: 1.7 mol CaCO3 × (3 mol O / 1 mol CaCO3) = 5.1 mol O
Converting Between Grams of Compound and Grams of Element
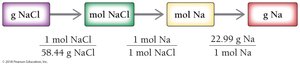
To find the mass of an element in a given mass of compound:
Step 1: Convert grams of compound to moles.
Step 2: Convert moles of compound to moles of element.
Step 3: Convert moles of element to grams.
Example: 15 g NaCl contains 5.9 g Na.
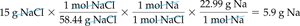
Mass Percent Composition
Definition and Calculation
Mass percent composition is the percentage by mass of each element in a compound. It is calculated as:
Example: Chromium in a metal oxide sample:
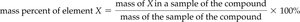
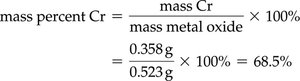
Using Mass Percent Composition as a Conversion Factor
Mass percent can be used to convert between grams of an element and grams of compound.
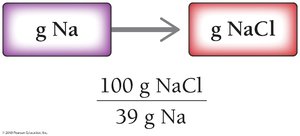
Example: Sodium chloride is 39% sodium by mass.
Conversion factor: 39 g Na : 100 g NaCl
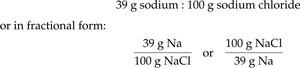
Application: Dietary Sodium
To stay within FDA guidelines for sodium intake, calculate the maximum grams of sodium chloride that can be consumed:
Given: 2.4 g Na, NaCl is 39% Na by mass.
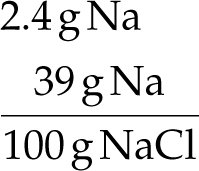
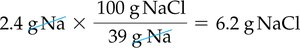
Mass Percent from Chemical Formula
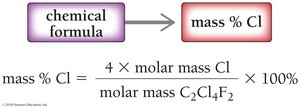
Mass percent can also be calculated from a chemical formula using molar masses:
Example: For C2Cl4F2, Cl mass percent = 69.58%

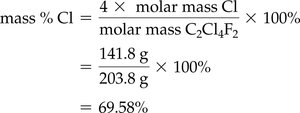
Empirical and Molecular Formulas
Empirical Formula
The empirical formula shows the simplest whole-number ratio of atoms in a compound. The molecular formula is a whole-number multiple of the empirical formula.
Example: Hydrogen peroxide (H2O2) has empirical formula HO.
Molecular formula = Empirical formula × n
Calculating Empirical Formula from Experimental Data
Steps to determine empirical formula:
Write down masses of each element.
Convert masses to moles using molar mass.
Write a pseudo-formula using mole values as subscripts.
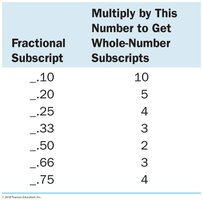
Divide all subscripts by the smallest value to get whole numbers.
If subscripts are not whole numbers, multiply by a small integer (see table below).

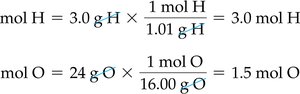
Calculating Molecular Formula from Empirical Formula and Molar Mass
The molecular formula is determined by multiplying the empirical formula by n, where:
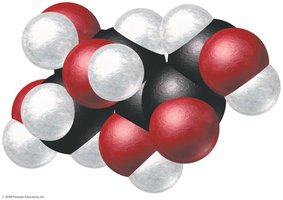
Example: For fructose, empirical formula CH2O, molar mass 180.2 g/mol, empirical formula molar mass 30.03 g/mol, n = 6.
Molecular formula = CH2O × 6 = C6H12O6
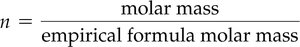
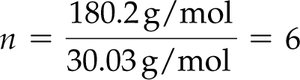
Summary and Learning Objectives
Key Concepts
The mole allows chemists to count atoms and molecules by weighing.
Molar mass relates mass to moles for elements and compounds.
Chemical formulas provide conversion factors for calculations.
Mass percent composition quantifies the proportion of elements in compounds.
Empirical and molecular formulas are determined from experimental data and molar mass.
Learning Objectives
Convert between moles and number of atoms.
Convert between grams and moles.
Convert between grams and number of atoms or molecules.
Convert between moles of a compound and moles of a constituent element.
Convert between grams of a compound and grams of a constituent element.
Use mass percent composition as a conversion factor.
Determine mass percent composition from a chemical formula.
Determine an empirical formula from experimental data.
Calculate a molecular formula from an empirical formula and molar mass.
