 Back
BackChapter 6: Language of Chemistry – Classification and Naming of Inorganic Compounds
Study Guide - Smart Notes

Language of Chemistry
Introduction to Chemical Nomenclature
The International Union of Pure and Applied Chemistry (IUPAC) sets the global standards for naming chemical compounds. Understanding these rules is essential for clear communication in chemistry, especially when classifying and naming inorganic compounds.
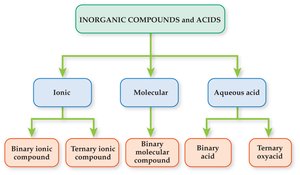
Classification of Inorganic Compounds
Main Classes of Inorganic Compounds
Most inorganic compounds do not contain carbon, with exceptions such as carbon dioxide (CO2) and carbonates (CO32–). Inorganic compounds are classified into five common types:
Binary ionic compounds
Ternary ionic compounds
Binary molecular compounds
Binary acids
Ternary oxyacids
Ionic Compounds
Ionic compounds are formed from the combination of metals and nonmetals. They are classified as:
Binary ionic compounds: Contain two elements (one metal, one nonmetal). Examples: NaCl, AlCl3.
Ternary ionic compounds: Contain three elements, often including oxygen. Examples: KNO3, Al(NO3)3.
Molecular Compounds
Binary molecular compounds consist of two nonmetal elements. Examples include ammonia (NH3) and water (H2O).
Aqueous Acids
Acids are classified based on their composition:
Binary acids: Aqueous solutions containing hydrogen and a nonmetal (e.g., HCl(aq)).
Ternary oxyacids: Aqueous solutions containing hydrogen, oxygen, and another nonmetal (e.g., HNO3(aq)).
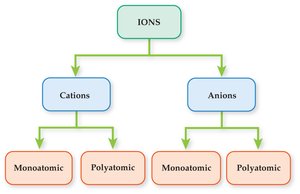
Classification of Ions
Types of Ions
An ion is an atom or group of atoms with a net electrical charge. Ions are classified as:
Cations: Positively charged ions (e.g., Na+, Mg2+).
Anions: Negatively charged ions (e.g., Cl–, SO42–).
Polyatomic ions: Groups of atoms with a charge (e.g., NO3–).
Monoatomic Ions
Monoatomic ions are single atoms with a charge. Metals lose electrons to form cations, named after the element (e.g., sodium ion, magnesium ion). Nonmetals gain electrons to form anions, named by adding the suffix -ide to the root name (e.g., chloride, oxide).
Charge Determination for Monoatomic Ions
Metal cations (A groups): Charge equals the family number on the periodic table.
Nonmetal anions (A groups): Charge equals the family number minus eight.
Exceptions: Tin and lead (Group IVA/14) form both 2+ and 4+ ions; many transition metals can have multiple charges.

Polyatomic Ions
Polyatomic ions are composed of multiple atoms, often including oxygen. Oxyanions are named with the suffix -ate or -ite:
-ate: Indicates more oxygen atoms (e.g., nitrate NO3–).
-ite: Indicates fewer oxygen atoms (e.g., nitrite NO2–).
-ide: Used for hydroxide (OH–) and cyanide (CN–).
Writing Chemical Formulas
Formulas of Ionic Compounds
An ionic compound is composed of positive and negative ions. The formula unit is neutral, so the total positive charge must equal the total negative charge:
If charges are equal: one of each ion (e.g., KBr, CaO).
If charges are not equal: use subscripts to balance charges (e.g., BaCl2, Li2S).
For polyatomic ions: use parentheses if more than one is needed (e.g., (NH4)2SO4).
Determining Ion Charges in Compounds
For metals with multiple possible charges, determine the charge based on the anion. Example: In Cr3N2, each Cr is 2+ because the total negative charge from N3– is 6–.
Naming Chemical Compounds
Naming Binary Ionic Compounds
Combine the cation and anion names (drop the word "ion"), with the cation first and the anion second. Add the suffix -ide to the anion.
Example: MgO is magnesium oxide.
Example: HgS is mercury(II) sulfide.

Predicting Formulas from Names
To determine the formula from the name, use the charges of the ions and balance them so the total charge is zero. Example: Iron(III) fluoride is FeF3 because Fe3+ and F– combine in a 1:3 ratio.
Naming Ternary Ionic Compounds
Name the cation followed by the anion. If the metal can have more than one oxidation state, indicate the charge with Roman numerals.
Example: KClO2 is potassium chlorite.
Example: Co(ClO3)3 is cobalt(III) chlorate.
Naming Binary Molecular Compounds
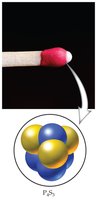
Binary molecular compounds are composed of two nonmetals. The first element is named first, and the second element has the suffix -ide. The number of atoms is indicated by Greek prefixes.
Example: P4S3 is tetraphosphorus trisulfide.
Exception: If there is only one atom of the first element, "mono-" is not used (e.g., CO is carbon monoxide).
Naming Acids
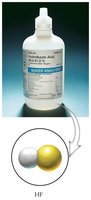
Binary Acids
Binary acids are aqueous solutions containing hydrogen and a nonmetal. The formula begins with H. Name by using the prefix hydro- and the suffix -ic acid.
Example: HF(aq) is hydrofluoric acid.
Example: HI(aq) is hydroiodic acid.

Ternary Oxyacids
Ternary oxyacids are aqueous solutions containing hydrogen and an oxyanion. If the acid is derived from an oxyanion ending in -ate, change the suffix to -ic acid. If derived from -ite, change the suffix to -ous acid.
Example: HNO3(aq) is nitric acid (from nitrate).
Example: HNO2(aq) is nitrous acid (from nitrite).
Summary Tables
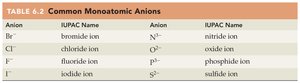
Common Monoatomic Anions
Anion | IUPAC Name | Anion | IUPAC Name |
|---|---|---|---|
Br– | bromide ion | N3– | nitride ion |
Cl– | chloride ion | O2– | oxide ion |
F– | fluoride ion | P3– | phosphide ion |
I– | iodide ion | S2– | sulfide ion |
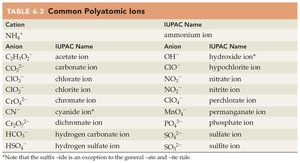
Common Polyatomic Ions
Cation | IUPAC Name | Anion | IUPAC Name |
|---|---|---|---|
NH4+ | ammonium ion | CH3COO– | acetate ion |
CO32– | carbonate ion | ||
ClO– | hypochlorite ion | ||
ClO2– | chlorite ion | ||
ClO3– | chlorate ion | ||
ClO4– | perchlorate ion | ||
CN– | cyanide ion | ||
HCO3– | hydrogen carbonate ion | ||
HSO4– | hydrogen sulfate ion | ||
OH– | hydroxide ion | ||
NO2– | nitrite ion | ||
NO3– | nitrate ion | ||
SO42– | sulfate ion | ||
SO32– | sulfite ion |
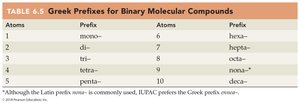
Greek Prefixes for Binary Molecular Compounds
Atoms | Prefix | Atoms | Prefix |
|---|---|---|---|
1 | mono- | 6 | hexa- |
2 | di- | 7 | hepta- |
3 | tri- | 8 | octa- |
4 | tetra- | 9 | nona-* |
5 | penta- | 10 | deca- |
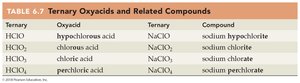
Ternary Oxyacids and Related Compounds
Ternary | Oxyacid | Ternary | Compound |
|---|---|---|---|
HClO | hypochlorous acid | NaClO | sodium hypochlorite |
HClO2 | chlorous acid | NaClO2 | sodium chlorite |
HClO3 | chloric acid | NaClO3 | sodium chlorate |
HClO4 | perchloric acid | NaClO4 | sodium perchlorate |
Summary
Most inorganic compounds do not contain carbon.
Binary ionic compounds: metal + nonmetal.
Ternary ionic compounds: metal + nonmetal + oxygen.
Binary molecular compounds: two nonmetals.
Acids: hydrogen + nonmetal or oxyanion.
Cations: element name + "ion"; charge indicated with Roman numerals if multiple possible charges.
Ionic compounds: cation first, anion second, anion ends with "-ide".
Molecular compounds: elements in order C, P, N, H, S, I, Br, Cl, O, F; Greek prefixes indicate atom count.
Acids: binary acids use "hydro-" and "-ic acid"; ternary oxyacids use "-ic acid" or "-ous acid" based on oxyanion suffix.
