 Back
BackChapter 7: Chemical Reactions – Study Guide and Key Concepts
Study Guide - Smart Notes

Chemical Reactions
Definition and Classes of Chemical Reactions
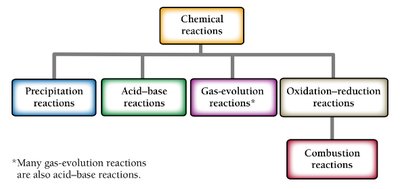
Chemical reactions involve the transformation of one or more substances into different substances. These processes are fundamental to chemistry and can be classified into several main types:
Precipitation reactions: Formation of a solid (precipitate) from aqueous solutions.
Acid-base (neutralization) reactions: Acid and base neutralize each other, often forming salt and water.
Gas-evolution reactions: Formation of a gas from a liquid reaction.
Oxidation-reduction (redox) reactions: Transfer of electrons between substances, including combustion reactions (reaction with oxygen producing heat and oxygen-containing products).
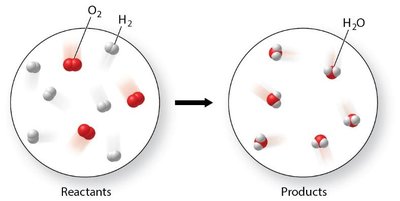
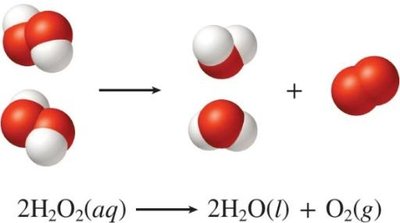
Example: The reaction of hydrogen and oxygen to form water illustrates the reorganization of atoms and the formation of new substances.
Evidence of Chemical Reactions
Not all chemical reactions result in observable changes, but common visible clues include:
Change in color
Formation of a solid
Formation of a gas
Production of light
Emission or absorption of heat










Chemical Equations
Structure and Balancing
Chemical equations are symbolic representations of chemical reactions. They show how atoms are reorganized, bonds are broken and formed, and new substances are produced. Mass is conserved in all chemical reactions, meaning atoms are neither created nor destroyed.
Reactants: Starting materials, written on the left side.
Products: New substances formed, written on the right side.
Phases: Physical state indicated in parentheses: (s) – solid, (l) – liquid, (g) – gas, (aq) – aqueous.
Example:
Balancing equations ensures mass conservation. Subscripts in chemical formulas cannot be changed; instead, coefficients are used to balance the number of atoms of each element.
Begin balancing with elements only contained in one reactant and one product.
Polyatomic ions can be treated as one unit if present on both sides.
Save oxygen and hydrogen for last.
Solutions and Solubility
Definitions and Properties
A solution is a homogeneous mixture of substances. The solute is the substance dissolved, usually in lesser amount, and the solvent is the substance doing the dissolving, usually in greater amount (often water, creating an aqueous solution).
Solubility & Dissociation of Ionic Compounds
Solubility is the maximum amount of solute that will dissolve in a given quantity of solvent at a specific temperature. Dissociation is the process by which an ionic compound breaks apart into its constituent ions upon dissolution. Water molecules surround each ion in a process called hydration.
Soluble ionic compounds dissociate completely.
Insoluble (slightly soluble) ionic compounds dissociate only partially.
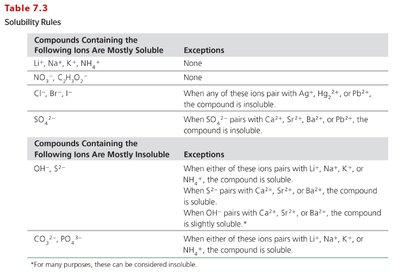
Solubility Rules
Solubility rules help predict whether an ionic compound will dissolve in water. Compounds containing certain ions are usually soluble, while others are mostly insoluble. Exceptions exist and must be checked for each rule.
Compounds Containing the Following Ions Are Mostly Soluble | Exceptions |
|---|---|
Li+, Na+, K+, NH4+ | None |
NO3-, C2H3O2- | None |
Cl-, Br-, I- | Ag+, Hg22+, Pb2+ |
SO42- | Ca2+, Sr2+, Ba2+, Pb2+ |
Compounds Containing the Following Ions Are Mostly Insoluble | Exceptions |
OH-, S2- | Li+, Na+, K+, NH4+, Ca2+, Sr2+, Ba2+ |
CO32-, PO43- | Li+, Na+, K+, NH4+ |
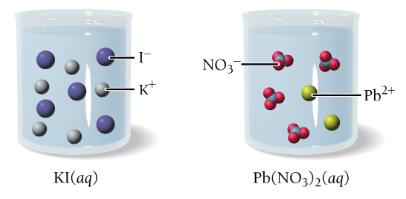
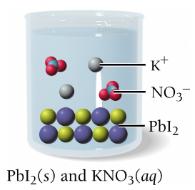
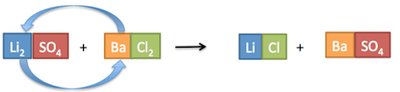
Precipitation Reactions
Mechanism and Prediction
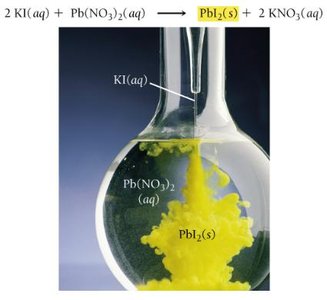
Precipitation reactions occur when two solutions containing soluble ionic compounds are mixed and an insoluble compound (precipitate) forms. The products are predicted by swapping ion partners, and solubility rules are used to identify the precipitate.
If no precipitate forms, no chemical reaction occurs.
Example: Mixing magnesium nitrate and potassium phosphate forms magnesium phosphate (precipitate) and potassium nitrate (aqueous).
Electrolytes
Types and Properties
An electrolyte is a substance that dissolves in water to yield a solution that conducts electricity. This occurs because the substance separates into ions, allowing electron flow.
Strong electrolytes: Dissociate completely; conduct current efficiently (soluble salts, strong acids, strong bases).
Weak electrolytes: Dissociate partially; conduct a small amount of current (insoluble salts, weak acids, weak bases).
Nonelectrolytes: Do not separate into ions; do not conduct current (neutral molecular compounds).

Chemical Equations: Molecular, Ionic, and Net Ionic
Types of Equations
Chemical reactions can be represented in three main ways:
Molecular equation: Substances written as neutral compounds.
Complete ionic equation: Substances written as they exist in solution (strong electrolytes as ions).
Net ionic equation: Only includes substances that participate in the reaction; spectator ions are removed.
Acid-Base Reactions (Neutralization)
Mechanism and Examples
Acid-base reactions involve an acid (producing H+ in solution) and a base (producing OH- in solution). The H+ associates with water to form H3O+ (hydronium ion). These reactions produce water and a salt.
Acids can be monoprotic, diprotic, or polyprotic (producing 1, 2, or more protons).
Bases can be monobasic, dibasic, or polybasic (producing 1, 2, or more OH-).
Example:
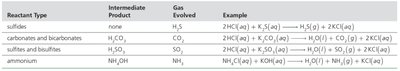
Gas-Evolution Reactions
Mechanism and Prediction
Gas-evolution reactions occur when two aqueous solutions mix to form a gaseous product. Some reactions form a gas directly, while others form an intermediate product that decomposes to produce a gas and water.
Reactant Type | Intermediate Product | Gas Evolved | Example |
|---|---|---|---|
Sulfides | none | H2S | 2HCl(aq) + K2S(aq) → H2S(g) + 2KCl(aq) |
Carbonates and bicarbonates | H2CO3 | CO2 | 2HCl(aq) + K2CO3(aq) → H2O(l) + CO2(g) + 2KCl(aq) |
Sulfites and bisulfites | H2SO3 | SO2 | 2HCl(aq) + K2SO3(aq) → H2O(l) + SO2(g) + 2KCl(aq) |
Ammonium | NH4OH | NH3 | NH4Cl(aq) + KOH(aq) → H2O(l) + NH3(g) + KCl(aq) |
Oxidation-Reduction (Redox) Reactions
Mechanism and Identification
Redox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons, and reduction is the gain of electrons. These processes always occur together.
LEO GER: Loss of Electrons is Oxidation, Gain of Electrons is Reduction.
OIL RIG: Oxidation Is Loss, Reduction Is Gain (of electrons).
Redox reactions are easily identified when:
A substance reacts with oxygen (combustion reaction).
A metal reacts with a nonmetal to form an ionic compound.
Atoms become ions or ions change charge.
General Types of Chemical Reactions
Classification and Generic Equations
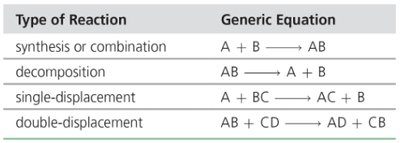
Chemical reactions can be classified into four main types:
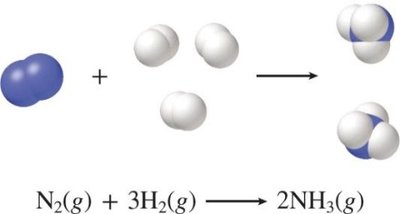
Combination (synthesis) reaction: Two or more substances combine to form one product. Generic:
Decomposition reaction: One substance breaks down into two or more products. Generic:
Single-replacement (displacement) reaction: A free element replaces another element in a compound. Generic:
Double-replacement (displacement) reaction: Two compounds exchange ions to form new compounds. Generic:
Chapter 7 Study Guide: Key Terms and Skills
Understand and use terms: chemical equation, reactants, products, phases, solution, solute, solvent, solubility, dissociation, hydration, soluble, insoluble, electrolyte, strong electrolyte, weak electrolyte, nonelectrolyte, precipitation reaction, precipitate, spectator ion, neutralization (acid-base) reaction, acid, base, hydronium ion, ionization, gas-evolution reaction, oxidation-reduction (redox) reaction, oxidation, reduction.
Know notation for indicating phases in chemical equations: (s), (l), (g), (aq).
Balance chemical equations and write equations from word descriptions.
Use solubility rules to predict phases of ionic compounds in reactions.
Distinguish strong, weak, and nonelectrolytes based on conductivity and compound type.
Predict products and phases for reactions between ionic compounds and acids/bases.
Recognize intermediate products for gas-evolution reactions and their decomposition.
Convert molecular equations to complete ionic and net ionic equations, identifying spectator ions.
Recognize types of reactions: precipitation, neutralization, gas-evolution, redox, combustion, combination, decomposition, single-replacement, double-replacement.
