 Back
BackChapter 7: Chemical Reactions – Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Chemical Reactions
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products) with different properties. These reactions are fundamental to both everyday life and industrial processes. For example, the reaction between baking soda and vinegar produces carbon dioxide gas, causing the classic 'volcano' eruption, while the combustion of hydrocarbons in car engines releases energy for movement.
Gas-evolution reactions: Reactions in liquids that produce a gas, such as the formation of CO2 in the baking soda and vinegar reaction.
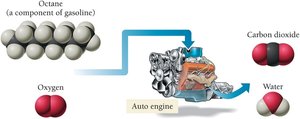
Combustion reactions: Reactions where a substance reacts with oxygen to produce heat, carbon dioxide, and water. These are a type of oxidation–reduction (redox) reaction.
Precipitation reactions: Reactions in which a solid forms from the mixing of two solutions, such as the removal of hard water ions in laundry detergents.

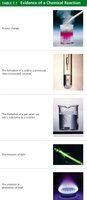
Evidence of a Chemical Reaction
Although we cannot directly observe atoms and molecules, chemical reactions often produce observable changes. These changes provide evidence that a chemical reaction has occurred.
Color change
Formation of a solid (precipitate) in a previously clear solution
Formation of a gas when a substance is added to a solution
Emission of light
Emission or absorption of heat
Note: Not all observable changes indicate a chemical reaction. For example, boiling water produces bubbles (gas), but no new substance is formed—this is a physical change, not a chemical reaction.
The Chemical Equation
Chemical equations are symbolic representations of chemical reactions. They show the reactants (on the left) and products (on the right), often including the physical state of each substance:
(s): solid
(l): liquid
(g): gas
(aq): aqueous (dissolved in water)
For example, the combustion of methane is represented as:
Balancing chemical equations ensures the law of conservation of mass is obeyed—atoms are neither created nor destroyed.
How to Write Balanced Chemical Equations
Balancing equations involves adjusting coefficients to ensure the same number of each atom on both sides:
Write correct formulas for all reactants and products.
Balance elements that appear in only one compound on each side first (usually metals before nonmetals).
Balance free elements last.
If necessary, multiply all coefficients to remove fractions.
Check that all atoms are balanced; only change coefficients, not subscripts.
Example: Reaction of aluminum with sulfuric acid:
Aqueous Solutions and Solubility
Terminology and Properties
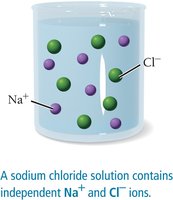
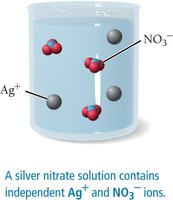
An aqueous solution is a homogeneous mixture of a substance dissolved in water. Compounds that dissolve are called soluble; those that do not are insoluble. When ionic compounds dissolve, they usually dissociate into ions, making the solution conductive if the compound is a strong electrolyte.
Strong electrolytes: Substances that completely dissociate into ions in solution (e.g., NaCl).
Non-electrolytes: Substances that do not produce ions in solution (e.g., pure water).
Solubility Rules
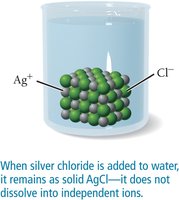
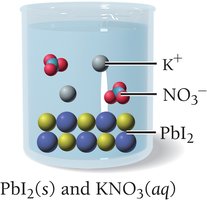
Solubility rules help predict whether an ionic compound will dissolve in water. For example, most compounds containing alkali metal ions or nitrate (NO3-) are soluble, while many silver, lead, and carbonate compounds are insoluble.
Soluble Compounds | Insoluble Compounds |
|---|---|
Alkali metal salts, NH4+, NO3-, C2H3O2- | AgCl, PbSO4, BaSO4, CaCO3 |
Additional info: See textbook for a complete solubility table.
Precipitation Reactions
Formation of a Precipitate
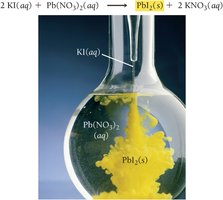
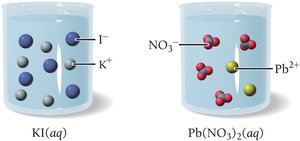
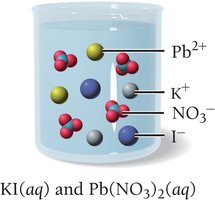
A precipitation reaction occurs when two aqueous solutions combine to form an insoluble solid (precipitate). Only insoluble compounds form precipitates. For example, mixing potassium iodide and lead(II) nitrate produces yellow lead(II) iodide:
When No Reaction Occurs
If all possible products are soluble, no precipitate forms and no reaction is observed. For example, mixing potassium iodide and sodium chloride results in no reaction because both possible products (KCl and NaI) are soluble.


Predicting and Writing Precipitation Reactions
Write correct formulas for reactants.
Combine cations and anions to predict possible products.
Use solubility rules to determine if any product is insoluble.
If a precipitate forms, write its formula with (s); otherwise, write 'No Reaction.'
Balance the equation by adjusting coefficients.
Oxidation–Reduction (Redox) Reactions
Definition and Examples
Redox reactions involve the transfer of electrons between substances. They are responsible for processes such as rusting, combustion, and battery operation. In redox reactions, one substance is oxidized (loses electrons) and another is reduced (gains electrons).
Oxidation: Loss of electrons
Reduction: Gain of electrons
Mnemonics: OIL RIG (Oxidation Is Loss, Reduction Is Gain); LEO GER (Lose Electrons Oxidation, Gain Electrons Reduction)

Examples of redox reactions:
Reaction with elemental oxygen
Reaction between a metal and a nonmetal
General electron transfer between substances
Combustion Reactions
Combustion reactions are a type of redox reaction where a substance reacts with oxygen to form one or more oxygen-containing compounds, often releasing heat. Hydrocarbons combust to form carbon dioxide and water:
Balance the equation by adjusting coefficients as needed.
Classifying Chemical Reactions
Types of Chemical Reactions
Chemical reactions can be classified based on the rearrangement of atoms:
Synthesis (Combination) Reaction: Two or more simple substances combine to form a more complex substance. Example:
Decomposition Reaction: A complex substance breaks down into simpler substances. Example:
Single-Displacement Reaction: One element displaces another in a compound. Example:
Double-Displacement Reaction: Elements or groups in two compounds exchange places. Example:

Double-displacement reactions include precipitation, acid–base, and gas evolution reactions.
Summary and Review
Chemical reactions involve the transformation of substances into new substances.
Evidence for chemical reactions includes color change, precipitate formation, gas evolution, light emission, and heat change.
Chemical equations must be balanced to reflect conservation of mass.
Aqueous solutions involve substances dissolved in water; solubility rules help predict precipitation.
Redox and combustion reactions involve electron transfer and energy release.
Reactions are classified as synthesis, decomposition, single-displacement, or double-displacement.
