 Back
BackCHEM 101 Exam III Review: Quantum Theory, Bonding, Gases, IMFs, and Solutions
Study Guide - Smart Notes

Quantum Mechanical Theory and Electron Configurations
Electron Configurations and Orbital Diagrams
Understanding electron configurations is essential for predicting chemical behavior. Electron configurations describe the arrangement of electrons in an atom's orbitals, following the Pauli exclusion principle and Hund's rule.
Neutral Atom: The number of electrons equals the atomic number (Z).
Filling Order: Electrons fill lower energy orbitals first (Aufbau principle), with a maximum of two electrons per orbital.
Hund's Rule: Half-fill all orbitals in a subshell before pairing electrons.
Valence Electrons: Electrons in the outermost shell (highest principal quantum number, n).
Noble Gas Notation: Use the symbol of the previous noble gas in brackets to abbreviate the configuration.
Example (Chlorine, Cl):
Atomic number: 17
Full configuration: 1s22s22p63s23p5
Noble gas notation: [Ne] 3s23p5
Valence configuration: 3s23p5
Unpaired electrons: 1
For ions: Add electrons for anions, remove for cations (remove from highest n first; for transition metals, remove s before d electrons).
Bohr Theory vs. Quantum Mechanical Theory
Atomic Structure Models
The Bohr model describes electrons in fixed orbits around the nucleus, while quantum mechanical theory uses orbitals—regions of probability where electrons are likely to be found.
Bohr Orbits: Fixed paths with quantized energies.
Quantum Orbitals: 3D regions (s, p, d, f) with specific shapes and orientations.
Absorption/Emission: Electrons absorb energy to move to higher energy levels (excited state) and emit energy when returning to lower levels (ground state).
Ground State: Lowest energy arrangement of electrons. Excited State: Any arrangement with higher energy than the ground state.
Electromagnetic Radiation (EMR) and the Spectrum
Relationships and Trends
Electromagnetic radiation includes all forms of light, characterized by energy (E), frequency (f), and wavelength (λ). These are related by:
Where is the speed of light, is Planck's constant. The visible spectrum ranges from red (longest wavelength, lowest energy) to violet (shortest wavelength, highest energy).
Periodic Trends
Atomic Size, Ionization Energy, and Metallic Character
Periodic trends help predict element properties:
Atomic Size: Increases down a group, decreases across a period.
Ionization Energy: Energy required to remove an electron; increases across a period, decreases down a group.
Metallic Character: Increases down a group, decreases across a period.
Chemical Bonding and Lewis Structures
Lewis Dot Structures
Lewis structures represent valence electrons as dots around element symbols, showing bonding and lone pairs.
Sum valence electrons for all atoms (adjust for ions).
Arrange atoms (central atom usually least electronegative).
Form bonds, complete octets (or duet for H), add lone pairs.
Check total electrons and octet rule.
Types of Bonding
Ionic Bonding: Transfer of electrons from metal to nonmetal, forming ions.
Covalent Bonding: Sharing of electrons between nonmetals.
VSEPR Theory and Molecular Shapes
Valence Shell Electron Pair Repulsion (VSEPR) theory predicts molecular shapes based on repulsion between electron groups around a central atom.
Count electron groups (bonds and lone pairs).
Determine electron geometry and molecular geometry.
Predict bond angles and molecular polarity.
# Electron Groups | Electron Geometry | Molecular Geometry | Bond Angles |
|---|---|---|---|
2 | Linear | Linear | 180° |
3 | Trigonal planar | Trigonal planar / Bent | 120° / <120° |
4 | Tetrahedral | Tetrahedral / Trigonal pyramidal / Bent | 109.5° / <109.5° |
Electronegativity and Bond Polarity
Electronegativity (EN) is an atom's ability to attract electrons in a bond. The difference in EN determines bond type:
ΔEN | Bond Type | Description |
|---|---|---|
< 0.5 | Nonpolar covalent | Electrons shared equally |
0.5 – 2.0 | Polar covalent | Electrons shared unequally (dipole) |
> 2.0 | Ionic | Electrons transferred |
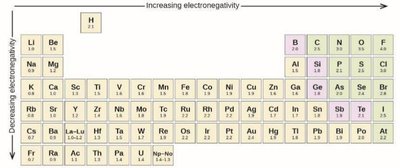
Molecular Polarity: Determined by both bond polarities and molecular shape (symmetry).
Gases and Gas Laws
Gas Laws and Calculations
Gas laws describe the relationships between pressure (P), volume (V), temperature (T), and amount (n) of a gas. Standard Temperature and Pressure (STP): 1 atm, 0°C (273.15 K).
Boyle's Law: (T, n constant)
Charles's Law: (P, n constant)
Avogadro's Law: (P, T constant)
Ideal Gas Law:
Always check units, especially for R (0.0821 L·atm/K·mol). Only use the ideal gas law for gases.

Intermolecular Forces (IMFs) and Physical Properties
Types of IMFs
London Dispersion: Present in all molecules; strength increases with size.
Dipole-Dipole: Present in polar molecules; positive end of one molecule attracts negative end of another.
Hydrogen Bonding: Strong dipole-dipole interaction between H and N, O, or F.
Physical properties such as boiling point (bp) and melting point (mp) are influenced by the strength of IMFs.
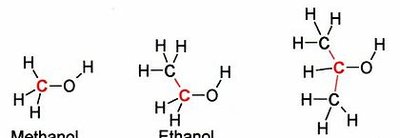
Example: Methanol, ethanol, and isopropanol all exhibit hydrogen bonding, but as molecular size increases, so do London forces, leading to higher boiling points.
Solutions and Concentration
Solution Terminology and Calculations
A solution is a homogeneous mixture of solute (minor component) and solvent (major component). Concentration can be expressed as:
Mass Percent:
Molarity (M):
Dilution Equation: (for dilution only)
"Like dissolves like": Polar solutes dissolve in polar solvents; nonpolar in nonpolar.
Stoichiometry with Solutions
Stoichiometry involves mole-to-mole conversions using balanced equations. For solutions, use molarity to convert between volume and moles. For gases, use the ideal gas law to find moles before stoichiometry.
Appendix: Useful Constants and Conversions
Quantity | Value |
|---|---|
1 atm | 760 mmHg = 760 torr = 101,325 Pa = 14.7 psi |
R (gas constant) | 0.0821 L·atm/K·mol |
K (Kelvin) | °C + 273.15 |
°C (Celsius) |
