 Back
BackLecture 9: Chemical Bonding: Atomic and Ionic Radii, Polyatomic Ions, and Lewis Structures
Study Guide - Smart Notes

Chemical Bonding
Introduction to Chemical Bonding
Chemical bonding describes the interactions that hold atoms together in compounds. Understanding how and why atoms bond is fundamental to chemistry, as it explains the structure and properties of matter. This section covers atomic and ionic radii, polyatomic ions, and the drawing of Lewis structures, all of which are essential for predicting and explaining chemical behavior.
Atomic and Ionic Radii
Atomic Radius
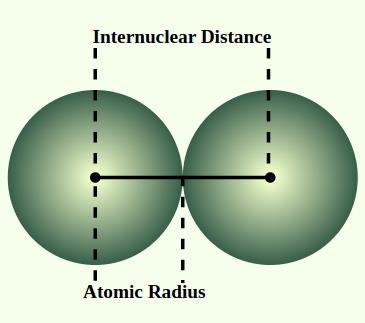
The atomic radius is defined as half the distance between the nuclei of two identical atoms bonded together. It provides insight into the size of an atom and influences how atoms interact in chemical reactions.
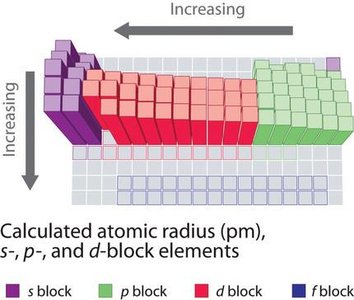
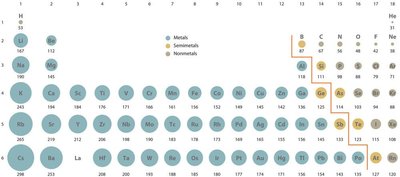
Trend Across a Period: Atomic radius decreases from left to right across a period due to increasing nuclear charge, which pulls electrons closer to the nucleus.
Trend Down a Group: Atomic radius increases down a group as additional electron shells are added, making atoms larger.
Importance: Atomic size affects bond lengths, reactivity, and the physical properties of elements and compounds.
Ionic Radius
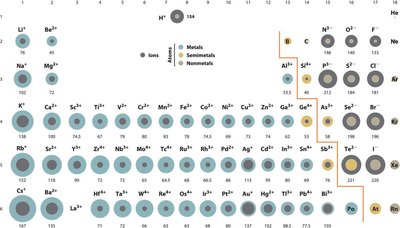
The ionic radius refers to the size of an ion. Cations (positively charged ions) are smaller than their parent atoms due to loss of electrons, while anions (negatively charged ions) are larger due to electron gain and increased electron-electron repulsion.
Cations: Smaller than neutral atoms; loss of electrons reduces electron-electron repulsion and allows the nucleus to pull remaining electrons closer.
Anions: Larger than neutral atoms; gain of electrons increases repulsion among electrons, expanding the ion.
Periodic Trends: Ionic radii follow similar trends to atomic radii but are also influenced by the charge and electron configuration of the ion.
Hydrated Ionic Radii
When ions are dissolved in water, they attract water molecules, forming a hydration shell. The hydrated ionic radius is the effective size of an ion surrounded by water molecules, which is important in biological and environmental chemistry.
Polyatomic Ions
Definition and Examples
Polyatomic ions are groups of covalently bonded atoms that carry an overall charge. They behave as single units in chemical reactions and are common in many compounds.
Examples:
Ammonium ion:
Sulfate ion:
Nitrate ion:
Phosphate ion:
Charge: The charge on a polyatomic ion results from the total number of electrons compared to the total number of protons in the group of atoms.
Charge | Name | Formula |
|---|---|---|
1+ | Ammonium | NH4+ |
1- | Nitrate | NO3- |
2- | Sulfate | SO42- |
3- | Phosphate | PO43- |
Naming Compounds with Polyatomic Ions
When naming compounds containing polyatomic ions, simply name the cation first and the anion second. Parentheses are used in chemical formulas when more than one polyatomic ion is present.
Example: is called ammonium sulfate.
Example: contains phosphate and hydroxide ions.
Lewis Structures and the HONC Rules
Valence Electrons and the Octet Rule
Lewis structures are diagrams that show the arrangement of valence electrons among atoms in a molecule. The octet rule states that atoms tend to form bonds to achieve eight electrons in their valence shell (except hydrogen, which seeks two).
Valence Electrons: Electrons in the outermost shell, involved in bonding.
Octet Rule: Atoms gain, lose, or share electrons to achieve a stable configuration of eight valence electrons.
The HONC Rules
The HONC rules summarize the typical number of bonds formed by hydrogen, oxygen, nitrogen, and carbon:
Hydrogen (H): 1 bond
Oxygen (O): 2 bonds
Nitrogen (N): 3 bonds
Carbon (C): 4 bonds
Steps for Drawing Lewis Structures
Follow these steps to draw Lewis structures for molecules and polyatomic ions:
Determine the total number of valence electrons (including any charge for ions).
Sketch a skeletal structure, placing the least electronegative atom (except hydrogen) in the center.
Place electrons as lone pairs around outer atoms to fulfill the octet rule.
Subtract the electrons used so far from the total number of valence electrons. Place any remaining electrons around the central atom.
If the central atom lacks an octet, move one or more lone pairs from an outer atom to form double or triple bonds as needed, following the HONC rules.
Examples: Diatomic Molecules and Compounds
O2 (Oxygen): Each oxygen atom has 6 valence electrons. The molecule forms a double bond to satisfy the octet rule for both atoms.
N2 (Nitrogen): Each nitrogen atom has 5 valence electrons. The molecule forms a triple bond to satisfy the octet rule for both atoms.
CO2 (Carbon Dioxide): Carbon forms two double bonds with two oxygen atoms, satisfying the octet rule for all atoms.
Lewis Structures for NO and NO2
Nitric oxide (NO) and nitrogen dioxide (NO2) are important atmospheric molecules. Their Lewis structures illustrate the use of single and double bonds, as well as the presence of unpaired electrons (free radicals in the case of NO).
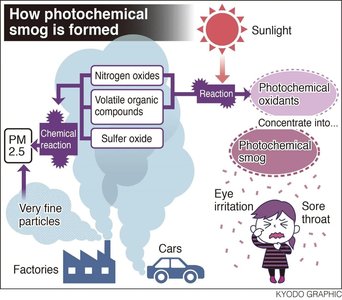
Free Radicals and Environmental Chemistry
Free Radicals
Free radicals are atoms or molecules with unpaired electrons, making them highly reactive. They play significant roles in atmospheric chemistry, pollution, and biological processes.
Example: Nitric oxide (NO) is a free radical involved in the formation of photochemical smog.
Example: Chlorofluorocarbons (CFCs) can generate free radicals that deplete the ozone layer.
Summary and Takeaways
Atomic and ionic radii influence chemical bonding and properties.
Polyatomic ions are common in chemistry and have specific naming conventions.
The HONC rules and Lewis structures help predict molecular structure and bonding.
Free radicals are important in environmental and atmospheric chemistry.
