 Back
Back2) Chemical Bonding: Ionic and Covalent Bonds, Electronegativity, and Polyatomic Ions
Study Guide - Smart Notes

Chemical Bonding
Introduction to Chemical Bonding
Chemical bonding is the process by which atoms combine to form compounds, resulting in the formation of molecules and extended structures. Understanding the types of bonds and the behavior of atoms is essential for predicting the properties and reactivity of substances in chemistry and the environment.
Ionic Compounds
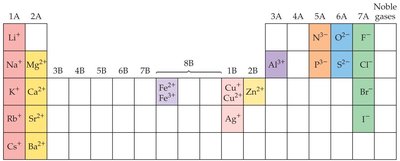
Formation of Ions
Atoms form ions by gaining or losing electrons to achieve a stable electron configuration, often resembling that of the nearest noble gas.
Metals lose electrons to form cations (positively charged ions).
Nonmetals gain electrons to form anions (negatively charged ions).
Ionic Bonds
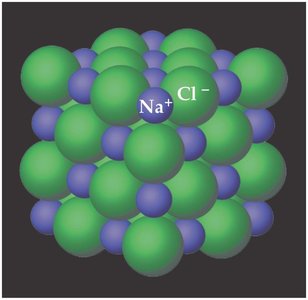
An ionic bond is the electrostatic attraction between oppositely charged ions in a compound. This type of bond typically forms between metals and nonmetals.
Example: Sodium chloride (NaCl) forms when sodium (Na+) and chloride (Cl-) ions attract each other.
Naming Ionic Compounds
The naming of ionic compounds follows specific rules:
The cation (metal) is named first, using the element's name.
The anion (nonmetal) is named by taking the root of the element's name and adding the suffix -ide.
For example, NaCl is sodium chloride, and MgO is magnesium oxide.
Formulas of Ionic Compounds
The chemical formula of an ionic compound reflects the ratio of ions needed to balance the overall charge to zero.
Example: The formula for calcium chloride is CaCl2 because two Cl- ions are needed to balance one Ca2+ ion.


Transition Metals and Variable Charges
Many transition metals can form more than one type of cation with different charges. The charge is indicated using Roman numerals in parentheses.
Example: Fe2+ is iron(II) ion, Fe3+ is iron(III) ion.
Example: Cu+ is copper(I) ion, Cu2+ is copper(II) ion.
Covalent Bonds
Formation of Covalent Bonds
Covalent bonds form when two nonmetal atoms share one or more pairs of electrons. This sharing allows each atom to achieve a stable electron configuration.
Single, double, and triple bonds correspond to the sharing of one, two, or three pairs of electrons, respectively.
Example: H2 (hydrogen gas) has a single covalent bond.
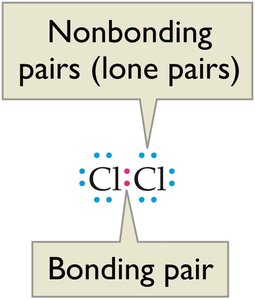
Bonding Pairs and Lone Pairs
In covalent molecules, electrons can be classified as bonding pairs (shared between atoms) or nonbonding pairs (lone pairs, not shared).
Example: In Cl2, each chlorine atom shares one pair (bonding pair) and has three lone pairs.
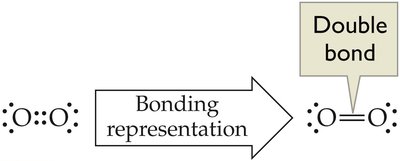
Double and Triple Bonds
Some atoms can share more than one pair of electrons, forming double or triple bonds.
Example: O2 (oxygen gas) has a double bond between the two oxygen atoms.
Electronegativity and Bond Polarity
Electronegativity
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. The greater the electronegativity, the more strongly an atom attracts bonding electrons.
Fluorine is the most electronegative element.
Electronegativity differences determine bond type and polarity.
Polar Covalent Bonds
When two atoms with different electronegativities form a bond, the shared electrons are drawn closer to the more electronegative atom, resulting in a polar covalent bond.
Example: In HCl, electrons are closer to Cl, making it partially negative (δ-) and H partially positive (δ+).
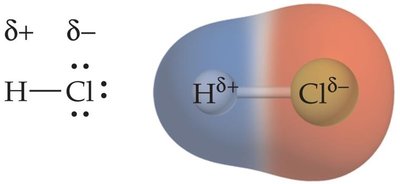
Bond Polarity and Electronegativity Difference
The difference in electronegativity between two atoms can be used to predict the type of bond:
< 0.5: Nonpolar covalent
0.5–2.0: Polar covalent
> 2.0: Ionic
HONC Rule
The HONC rule summarizes the typical bonding patterns of common elements:
Hydrogen forms 1 bond
Oxygen forms 2 bonds
Nitrogen forms 3 bonds
Carbon forms 4 bonds
Polyatomic Ions
Definition and Examples
Polyatomic ions are groups of covalently bonded atoms that carry a net charge. They behave as single units in chemical reactions and compounds.
Charge | Name | Formula |
|---|---|---|
1+ | Ammonium ion | NH4+ |
1+ | Hydronium ion | H3O+ |
1− | Nitrate ion | NO3− |
2− | Sulfate ion | SO42− |
3− | Phosphate ion | PO43− |
1− | Hydroxide ion | OH− |
1− | Acetate ion | CH3COO− |
Naming Compounds with Polyatomic Ions
When naming compounds containing polyatomic ions, simply name the cation and the polyatomic ion in order.
Example: NH4NO3 is ammonium nitrate.
Example: Ca5(PO4)3(OH) is hydroxyapatite.
Summary and Takeaways
Identify and describe ionic and covalent compounds.
Understand the role of electronegativity in bond formation and polarity.
Recognize and name common polyatomic ions and their compounds.
