 Back
BackLecture 10: Chemical Bonding, Lewis Structures, and Molecular Shapes: Introductory Chemistry Study Notes
Study Guide - Smart Notes

Review: Chemical Bonding & Lewis Structures
Introduction to Chemical Bonding
Chemical bonding is the process by which atoms combine to form compounds through the sharing or transfer of electrons. Understanding how atoms bond and the shapes of resulting molecules is fundamental to chemistry.
Ionic bonds occur when electrons are transferred from one atom to another, typically between metals and nonmetals.
Covalent bonds involve the sharing of electrons between nonmetal atoms.
Lewis structures are diagrams that show the bonding between atoms and the arrangement of valence electrons.
Cr2O3 is balances because Cr has 6 valence electrons and O also has 6 valence electrons. One has cation and the other an anion
Electron Shielding and Atomic Properties
Electron Shielding
Electron shielding refers to the reduction in effective nuclear charge on the valence electrons due to the presence of inner electrons. This concept helps explain trends in atomic size and reactivity across the periodic table.
Effective nuclear charge (Zeff) is the net positive charge experienced by valence electrons.
Inner electrons shield outer electrons from the full charge of the nucleus.
Electron shielding increases down a group, leading to larger atomic radii.
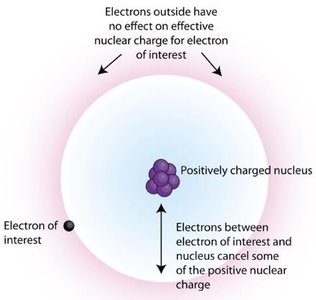
an electron in the outside had a pull with the proton in the nucleus, but some electrons are closer to the nucleus, essentially blocking/shielding/interfering with that electron's pull.
Atomic Radius and Periodic Trends
The atomic radius is the distance from the nucleus to the outermost electron shell. It varies predictably across the periodic table due to electron shielding and effective nuclear charge.
Atomic radius increases down a group (more electron shells).
Atomic radius decreases across a period (greater nuclear charge pulls electrons closer).
Periodic trends are visualized using block diagrams and periodic tables.
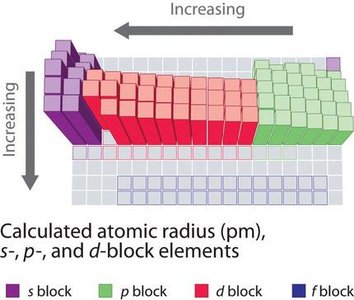
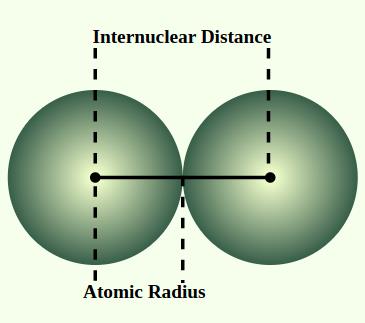
largest side in the bottom left
Electronegativity
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. It is highest for fluorine and decreases down a group and across a period from right to left.
Electronegativity affects bond polarity and molecular properties.
Periodic tables can display electronegativity values for each element.
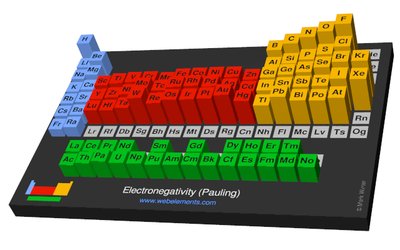
oxygen and fluorine are the most electronegative because they are small and really want an extra electron
cations have quick to ditch their electrons while the ones of the right don't want to let go of their electrons that's why they are mostly on the receiving end
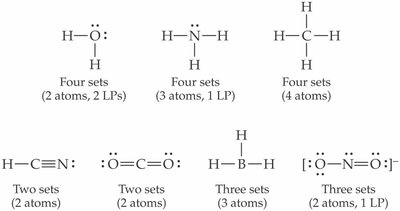
Lewis Structures
Drawing Lewis Structures for Diatomic Molecules
Lewis structures represent the arrangement of valence electrons in molecules. For diatomic molecules like O2 and N2, follow these steps:
Determine the total number of valence electrons available (including charge for ions).
Determine the total number of electrons needed by all atoms (usually 8 per atom for the octet rule).
Subtract the number of electrons you have from the number you need to find the number of bonding electrons.
Divide by 2 to get the number of bonds.
Fill in outer, lone pair electrons to ensure all atoms have 8 electrons.
Drawing Lewis Structures for Compounds
For more complex compounds, the process is expanded:
Count valence electrons.
Sketch a skeletal structure (central atom is usually the least electronegative, non-hydrogen atom).
Place electrons as lone pairs around outer atoms to fulfill the octet rule.
Subtract electrons used so far from the total number of valence electrons available. Place any remaining electrons around the central atom.
If the central atom lacks an octet, move lone pairs from outer atoms to form double or triple bonds as needed.
Follow HONC rules for maximum number of bonds (Hydrogen: 1, Oxygen: 2, Nitrogen: 3, Carbon: 4).
Lewis Structures: NOx Pollution (NO, NO2)
Nitrogen oxides (NO and NO2) are important atmospheric pollutants. Their Lewis structures help understand their reactivity and environmental impact.
NO: Nitric oxide has 11 valence electrons, resulting in an odd-electron molecule.
NO2: Nitrogen dioxide has 17 valence electrons, and its structure includes a lone electron.
Sources include cars, factories, and fertilized croplands.

Molecular Shapes and VSEPR Theory
Valence-Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the three-dimensional shapes of molecules based on the repulsion between electron pairs around a central atom. The arrangement of electron sets determines the molecular geometry.
Electron sets include both bonding pairs and lone pairs.
Shapes are determined by minimizing repulsion, placing electron sets as far apart as possible.
Common shapes: linear, trigonal planar, tetrahedral, trigonal pyramidal, bent.
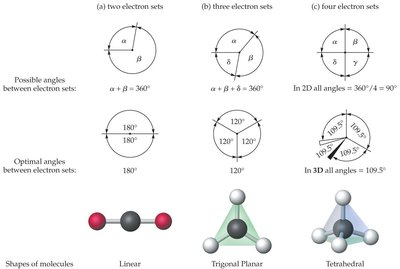
Molecular Shape | Bonded Atoms | Lone Pairs | Examples |
|---|---|---|---|
Linear | 2 | 0 | CO2, HCN |
Trigonal Planar | 3 | 0 | BF3, CH2O |
Tetrahedral | 4 | 0 | CH4, SiCl4 |
Trigonal Pyramidal | 3 | 1 | NH3, PCl3 |
Bent | 2 | 2 | H2O, H2S |
Bent | 2 | 1 | SO2, O3 |
Determining Molecular Shape
To determine the shape of a molecule or polyatomic ion:
Draw a Lewis structure, including bonds and lone pairs.
Count the number of electron sets around the central atom (multiple bonds count as one set).
Arrange electron sets as far apart as possible.
If there are no lone pairs, the arrangement is the molecular shape. If lone pairs are present, remove them to see the shape.
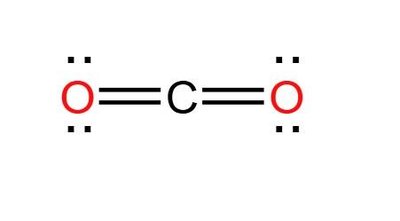
Examples: CO2 and H2O
Carbon dioxide (CO2) and water (H2O) are classic examples for illustrating molecular shapes:
CO2: Linear shape, two electron sets, bond angle 180°.
H2O: Bent shape, four electron sets (two bonds, two lone pairs), bond angle ~104.5°.
Summary and Key Takeaways
Summary of Concepts
Electron shielding affects atomic size and reactivity.
Lewis structures are essential for visualizing bonding and electron arrangement.
VSEPR theory predicts molecular shapes based on electron pair repulsion.
Understanding molecular geometry is crucial for predicting chemical behavior.
each bond has 2 electrons. in a double bond=4 electrons, triple bond= 6 electrons. each bond is a lone pair
each electron set is as far away from each other
Additional Resources
Periodic table visualizations: webelements.com
