 Back
BackChemical Bonding: Lewis Structures, Molecular Geometry, and Electronegativity
Study Guide - Smart Notes

Chemical Bonding Theories
Introduction to Bonding Theories
Bonding theories are essential for understanding how atoms combine to form compounds and why certain combinations are stable while others are not. These theories also explain the shapes of molecules, which influence their physical and chemical properties.
Bonding theories predict the types of compounds that can form and their molecular structures.
They explain why compounds like NaCl and H2O have their specific formulas.
Lewis Theory of Bonding
The Lewis theory, developed by G. N. Lewis, uses dot structures to represent valence electrons in atoms and molecules. These Lewis structures help predict molecular stability and geometry.
Electrons are represented as dots around the element symbol.
The number of valence electrons for main-group elements equals the group number (except helium, which has 2).
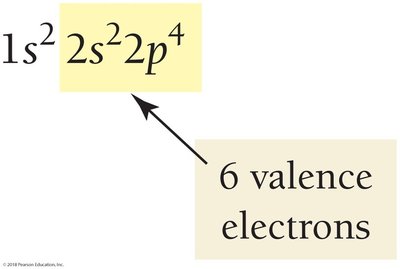

Lewis Structures for Period 2 Elements
Lewis structures visually display the number of valence electrons, making it easy to identify atoms with a stable octet (8 electrons).
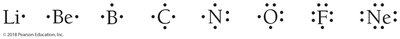
Writing Lewis Structures for Elements
To write a Lewis structure, place dots around the element symbol to represent valence electrons. For example, phosphorus (Group 5A) has 5 valence electrons.

Exception: Helium
Helium is stable with only two valence electrons (a duet), not an octet.

Types of Chemical Bonds
Ionic and Covalent Bonds
In Lewis theory, chemical bonds form by the transfer or sharing of electrons to achieve stable electron configurations:
Ionic bonds: Electrons are transferred from one atom to another, forming cations and anions.
Covalent bonds: Electrons are shared between atoms.
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight electrons in their valence shell.
Lewis Structures of Ionic Compounds
For example, potassium (K) transfers its valence electron to chlorine (Cl), forming K+ and Cl− ions.

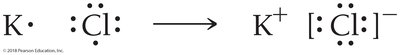
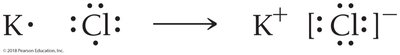
Predicting Chemical Formulas with Lewis Theory
Lewis structures help determine the correct ratios of ions in compounds. For sodium and sulfur, two sodium atoms are needed for each sulfur atom to satisfy the octet rule, resulting in Na2S.
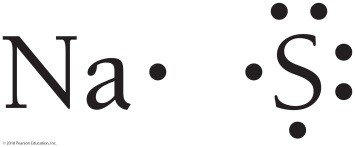
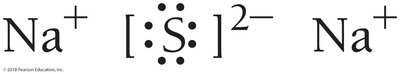
Lewis Structures for Covalent Compounds
Steps for Writing Lewis Structures
Write the skeletal structure (hydrogen is always terminal; symmetry is common).
Sum the valence electrons for all atoms (adjust for ion charge).
Distribute electrons to give octets (or duets for hydrogen), starting with bonding pairs, then lone pairs.
Covalent Lewis Structures: Electrons Shared
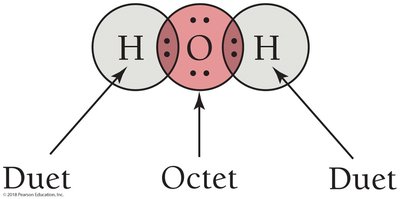
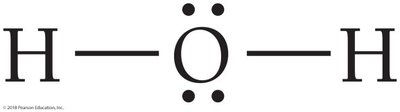
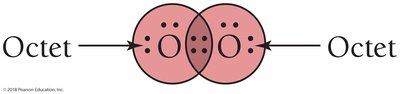
In water (H2O), hydrogen and oxygen share electrons so that hydrogen achieves a duet and oxygen an octet.


Bonding and Lone Pairs
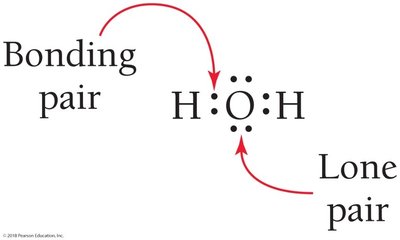
Shared electrons between atoms are bonding pairs; electrons localized on one atom are lone pairs. Bonding pairs are often shown as dashes.
Diatomic Molecules and the Octet Rule
Halogens (e.g., Cl2) and hydrogen (H2) form diatomic molecules to achieve stable electron configurations.

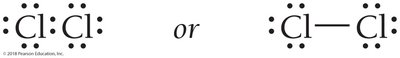
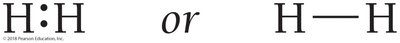
Double and Triple Bonds
Atoms can share more than one pair of electrons to achieve octets. Oxygen forms a double bond in O2, and nitrogen forms a triple bond in N2.


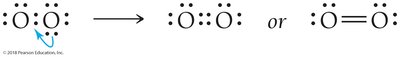


Lewis Structures for Polyatomic Ions
For polyatomic ions, adjust the total electron count for the ion's charge and enclose the structure in brackets with the charge indicated.
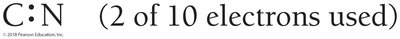
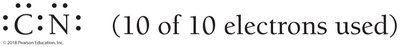
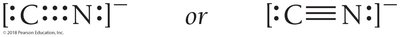
Exceptions to the Octet Rule
Duet rule: H, Li, and Be are stable with two electrons.
Incomplete octet: Al and B are stable with six electrons.
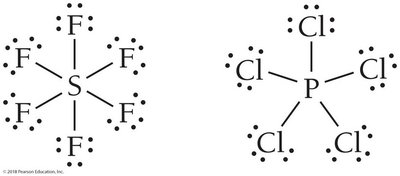
Expanded octet: Elements like Br, Xe, P, and Cl can have more than eight electrons.
Odd electron compounds: Some molecules (e.g., NO) have an odd number of electrons.
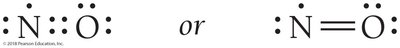
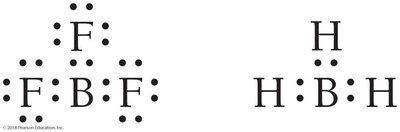
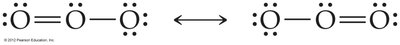
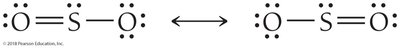
Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are multiple valid Lewis structures for the same molecule, indicating delocalized electrons. The true structure is an average of all resonance forms.
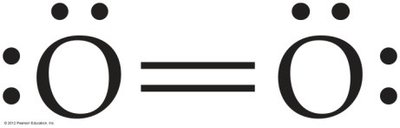
Molecular Geometry and VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts molecular shapes based on the repulsion between electron groups (lone pairs, single, double, or triple bonds) around a central atom. Electron groups arrange themselves to minimize repulsion, determining the molecule's geometry.
Examples of Molecular Geometry
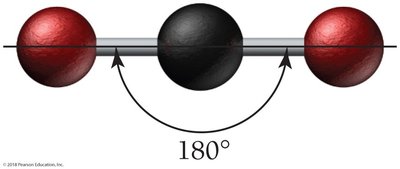
CO2: Linear geometry, bond angle 180°.

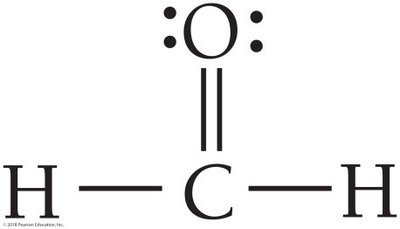
H2CO: Trigonal planar geometry, bond angles ~120°.
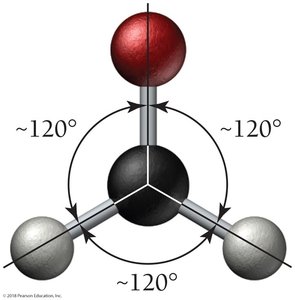
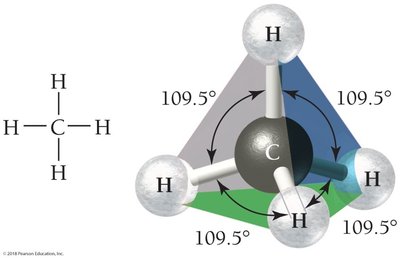
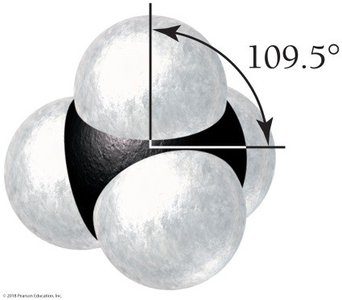
CH4: Tetrahedral geometry, bond angles 109.5°.
Molecules with Lone Pairs
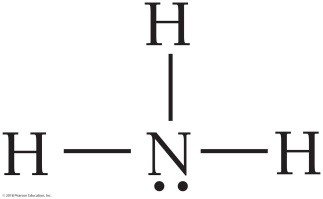
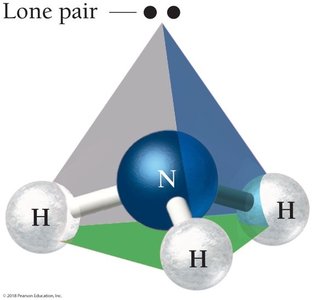
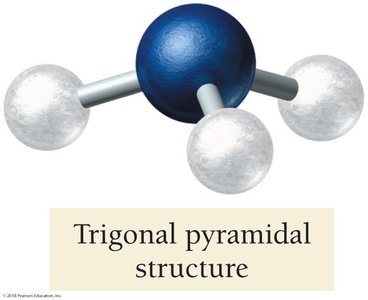
NH3: Electron geometry is tetrahedral, but molecular geometry is trigonal pyramidal due to one lone pair.
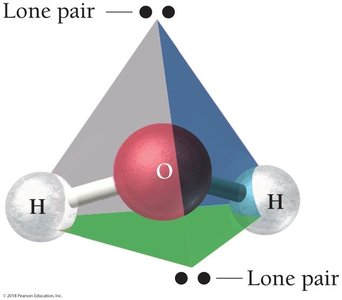
H2O: Electron geometry is tetrahedral, but molecular geometry is bent due to two lone pairs.

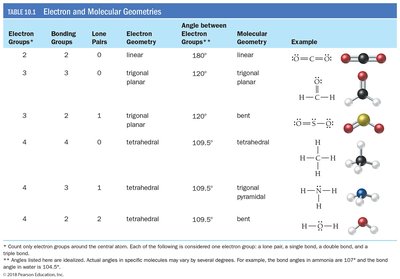
Summary Table: Electron and Molecular Geometries
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Angle | Molecular Geometry | Example |
|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | 180° | Linear | CO2 |
3 | 3 | 0 | Trigonal planar | 120° | Trigonal planar | H2CO |
3 | 2 | 1 | Trigonal planar | 120° | Bent | SO2 |
4 | 4 | 0 | Tetrahedral | 109.5° | Tetrahedral | CH4 |
4 | 3 | 1 | Tetrahedral | 109.5° | Trigonal pyramidal | NH3 |
4 | 2 | 2 | Tetrahedral | 109.5° | Bent | H2O |
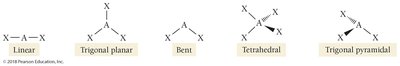
Representing Molecular Geometries on Paper
Chemists use different line notations to represent three-dimensional structures on two-dimensional paper:
Straight line: Bond in the plane of the paper
Hashed lines: Bond projecting into the paper
Wedge: Bond projecting out of the paper

Electronegativity and Polarity
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a covalent bond. Fluorine is the most electronegative element (4.0 on the Pauling scale).
Electronegativity increases across a period and decreases down a group in the periodic table.
Polar Covalent Bonds
When two atoms with different electronegativities share electrons, the electrons are not shared equally, resulting in a polar covalent bond with a dipole moment (separation of charge).
The more electronegative atom gains a partial negative charge (δ−), and the less electronegative atom gains a partial positive charge (δ+).
Bond Polarity and Molecular Polarity
If two atoms have identical electronegativities, the bond is nonpolar covalent (e.g., Cl2).
A large electronegativity difference (metal and nonmetal) leads to ionic bonds (e.g., NaCl).
An intermediate difference (two different nonmetals) leads to polar covalent bonds (e.g., HF).
Polar Molecules
A molecule is polar if its polar bonds do not cancel out, resulting in a net dipole moment. For example, water (H2O) is polar, while carbon dioxide (CO2) is nonpolar due to its linear geometry causing dipole moments to cancel.
Why Oil and Water Don’t Mix
Polar molecules (like water) interact strongly with each other, excluding nonpolar molecules (like oil), leading to phase separation.
Chapter Summary
Lewis theory: Atoms transfer or share electrons to achieve noble gas configurations (octet or duet).
Molecular shapes: Predicted by VSEPR theory based on electron group repulsion.
Electronegativity: Determines bond polarity and molecular polarity.
Polarity: Polar bonds and molecular geometry together determine if a molecule is polar or nonpolar.
