 Back
BackChemical Bonding: Lewis Structures, Molecular Geometry, and Polarity
Study Guide - Smart Notes

Chemical Bonding
Introduction to Chemical Bonding
Chemical bonding describes how atoms combine to form compounds, influencing the properties and behavior of substances. Understanding bonding is essential for predicting molecular structure, reactivity, and physical properties.
Chemical bonds are formed by the transfer or sharing of electrons between atoms.
Bonding theories, such as the Lewis model, help predict which combinations of atoms form stable compounds.
Bonding models are crucial in drug design and understanding biological molecules.
Lewis Model of Chemical Bonding
Valence Electrons and Lewis Structures
The Lewis model represents valence electrons as dots around an element's symbol, providing a visual method to predict bonding and molecular stability.
Valence electrons are the electrons in the outermost shell of an atom and are involved in bonding.
The number of valence electrons for main-group elements equals the group number (except helium).
Lewis structures (dot structures) show how atoms achieve stable electron configurations by sharing or transferring electrons.
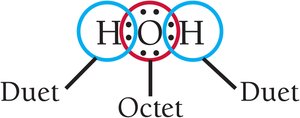
Atoms with eight valence electrons (an octet) are particularly stable; helium is an exception, stable with two electrons (a duet).
Types of Chemical Bonds
Atoms bond to achieve stable electron configurations, either by transferring or sharing electrons.
Ionic bonds: Electrons are transferred from a metal to a nonmetal, forming cations and anions.
Covalent bonds: Electrons are shared between nonmetals, forming molecules.
Octet rule: Atoms tend to gain, lose, or share electrons to achieve eight in their valence shell (except H and He).
Lewis Structures for Ionic Compounds
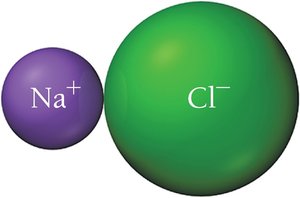
In ionic compounds, electrons are transferred from metals to nonmetals, resulting in charged ions held together by electrostatic attraction.
Example: Formation of KCl involves potassium transferring an electron to chlorine, forming K+ and Cl-.
Lewis structures for ions are shown with brackets and charges.
Stoichiometry is determined by the number of electrons transferred (e.g., Na2S requires two Na atoms for one S atom).
Lewis Structures for Covalent Compounds
Covalent compounds form when nonmetals share electrons to achieve octets or duets.
Shared electrons are called bonding pairs; unshared electrons are lone pairs.
Bonding pairs are often represented by dashes in Lewis structures.
Some elements form double or triple bonds to satisfy the octet rule.
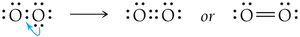

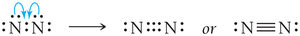
Steps to Write Lewis Structures for Covalent Compounds
Follow these steps to construct accurate Lewis structures:
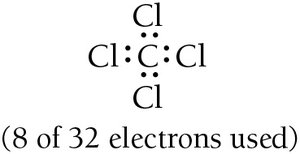
Write the correct skeletal structure, placing the least electronegative atom in the center (except hydrogen).
Calculate the total number of valence electrons, adjusting for ionic charges if necessary.
Distribute electrons to form bonds and complete octets (or duets for hydrogen).
If necessary, convert lone pairs to bonding pairs to satisfy the octet rule.

Lewis Structures for Polyatomic Ions
Polyatomic ions require special attention to charge when calculating total electrons. Their Lewis structures are enclosed in brackets with the charge indicated.
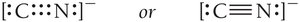
Exceptions to the Octet Rule
Some molecules do not follow the octet rule:
Odd-electron molecules (free radicals) cannot have all atoms with octets.
Boron compounds often have only six electrons around boron.
Expanded octets occur in some molecules with more than eight electrons around the central atom (not covered in detail).
Resonance Structures
Some molecules can be represented by more than one valid Lewis structure. These are called resonance structures, and the true structure is an average of all possibilities.
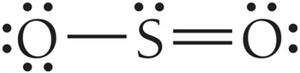
Molecular Geometry and VSEPR Theory
Predicting Molecular Shapes
The shape of a molecule is determined by the repulsion between electron groups around the central atom, as described by Valence Shell Electron Pair Repulsion (VSEPR) theory.
Electron geometry considers all electron groups (bonds and lone pairs).
Molecular geometry considers only the arrangement of atoms.
Bond angles depend on the number and type of electron groups.
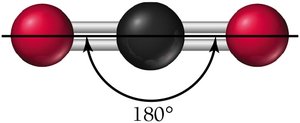
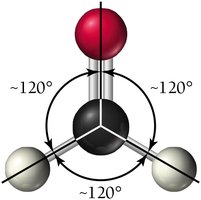
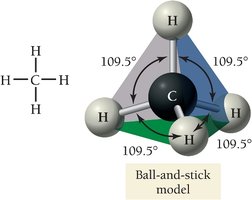
Molecular Geometry with Lone Pairs
Lone pairs on the central atom affect molecular geometry and bond angles.
NH3 has a trigonal pyramidal geometry due to one lone pair.
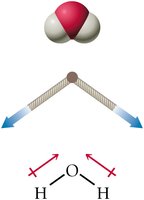
H2O has a bent geometry due to two lone pairs.
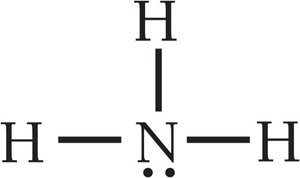
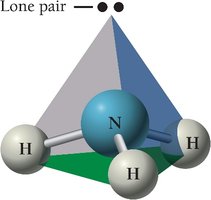
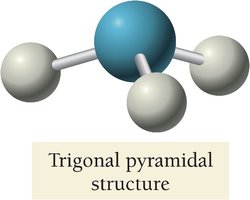

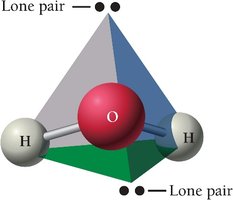
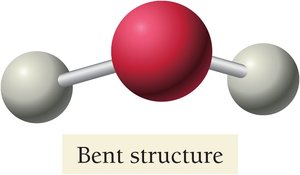
Summary Table: Electron and Molecular Geometries
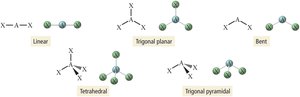
Electron and molecular geometries can be summarized as follows:
Electron Groups | Electron Geometry | Molecular Geometry | Bond Angles |
|---|---|---|---|
2 | Linear | Linear | 180° |
3 | Trigonal planar | Trigonal planar / Bent | 120° |
4 | Tetrahedral | Tetrahedral / Trigonal pyramidal / Bent | 109.5° |
Additional info: | Actual bond angles may vary due to lone pairs. |
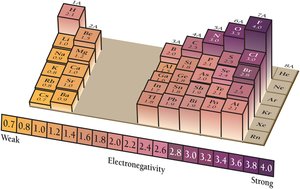
Electronegativity and Polarity
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a covalent bond. It determines bond polarity and influences molecular interactions.
Electronegativity increases across a period and decreases down a group.
Fluorine is the most electronegative element (4.0 on the Pauling scale).
Bond Polarity and Dipole Moments
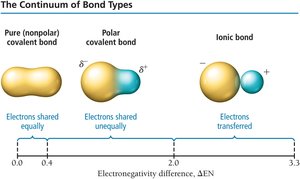
The difference in electronegativity between two atoms determines the type of bond:
Nonpolar covalent bond: Electrons shared equally (e.g., Cl2).
Polar covalent bond: Electrons shared unequally, creating partial charges and a dipole moment (e.g., HCl).
Ionic bond: Electrons transferred completely (e.g., NaCl).

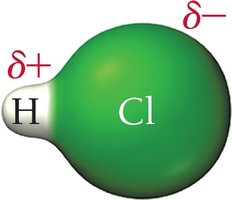
Polar Molecules
A molecule is polar if its polar bonds do not cancel out, resulting in a net dipole moment. Molecular geometry plays a crucial role in determining overall polarity.
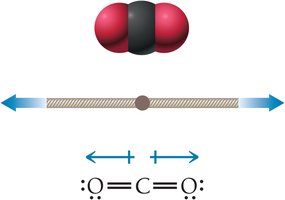
CO2 is nonpolar despite having polar bonds due to its linear geometry.
H2O is polar because its bent geometry prevents dipole cancellation.
Applications and Everyday Chemistry
Why Oil and Water Don’t Mix
Water molecules are polar, while oil molecules are nonpolar. Polar molecules attract each other and exclude nonpolar molecules, leading to phase separation.
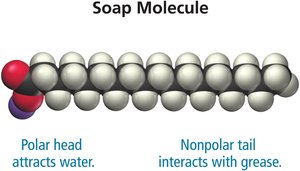
How Soap Works
Soap molecules have a polar head and a nonpolar tail, allowing them to interact with both water and grease, enabling the removal of nonpolar substances from surfaces.
Review and Learning Objectives
Write Lewis structures for elements, ionic, and covalent compounds.
Determine whether a compound is ionic or covalent.
Write resonance structures.
Predict electron and molecular geometry using VSEPR theory.
Determine molecular polarity based on bond polarity and geometry.
