 Back
BackChemical Kinetics: Collision Theory and Factors Affecting Reaction Rates
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed (rate) at which chemical reactions occur and the factors that influence these rates. Understanding kinetics is essential for controlling reactions in industrial, biological, and environmental contexts.
Collision Theory
Basic Principles of Collision Theory
Collision theory explains how and why chemical reactions occur. For a reaction to take place, reactant particles must collide. However, not all collisions result in a reaction; only those with proper orientation and sufficient energy can lead to product formation.
Collision Requirement: Reacting species must physically collide.
Orientation Requirement: The specific atoms or active sites must align correctly during the collision.
Energy Requirement: Colliding particles must possess enough energy to overcome the activation energy barrier.
Activation Energy (Ea): The minimum energy required for a reaction to occur.

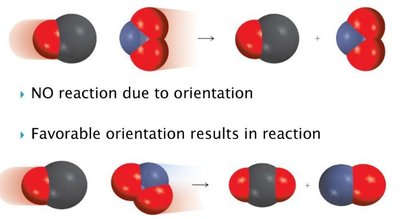
Complex Molecules and Orientation
For complex molecules, the orientation of the collision is critical. Only collisions where the correct atoms or functional groups meet will result in a reaction. This makes reactions involving large or complex molecules less likely to occur spontaneously.
Example: In organic reactions, only specific atoms must interact for bond formation.
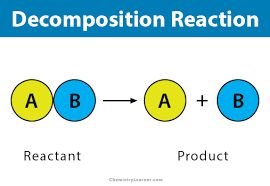
Decomposition Reactions
Decomposition reactions involve a single species breaking apart into two or more products. These reactions are simpler because collision orientation is not a factor; the process depends mainly on the energy supplied to the molecule.
Example:
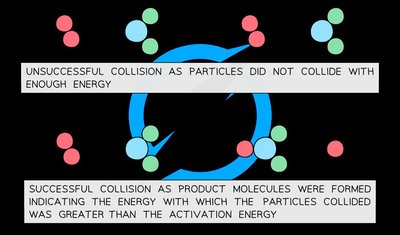
Activation Energy and Energy Barrier
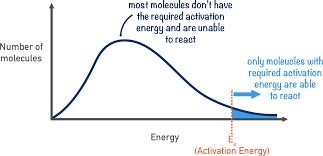
Activation energy is the energy threshold that must be exceeded for reactants to transform into products. Gentle collisions lacking sufficient energy will not result in a reaction. Energy profiles illustrate the activation energy as a 'hump' that must be overcome.
Bond Breaking: Requires energy input (activation energy).
Bond Making: Releases energy.
Factors Affecting Reaction Rates
Overview of Factors
Several factors influence the rate of a chemical reaction by affecting collision frequency, orientation, and energy:
Temperature
Surface Area
Concentration
Catalysts
Temperature
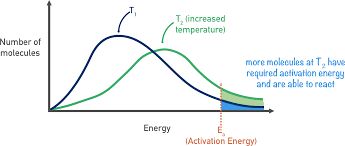
Temperature is a measure of the average kinetic energy of particles. Increasing temperature causes particles to move faster, increasing both the frequency and energy of collisions. More particles will have energy equal to or greater than the activation energy, leading to a higher reaction rate.
Higher Temperature: More frequent and energetic collisions.
Maxwell-Boltzmann Distribution: Shows how temperature affects the number of particles with sufficient energy to react.
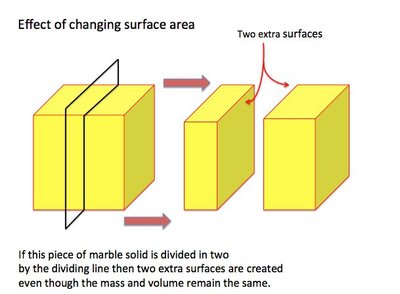
Surface Area
When a solid reacts with a liquid or gas, only the particles on the surface can participate in collisions. Increasing the surface area exposes more particles, increasing the frequency of collisions and thus the reaction rate.
Breaking solids: Dividing a solid into smaller pieces increases surface area.
Shape changes: Altering the shape of a solid can expose more surface area.
Exception: If a powder is too tightly packed, its effective surface area may decrease.

Concentration
Concentration refers to the number of particles in a given volume. Higher concentration increases the likelihood of collisions, leading to a faster reaction rate. In decomposition reactions, doubling the concentration doubles the number of particles available to react.
Single Particle: More particles in a space increases collision frequency.
Two-Particle Collisions: Higher concentration makes collisions more likely.
Rate Law: For many reactions, rate is proportional to concentration.

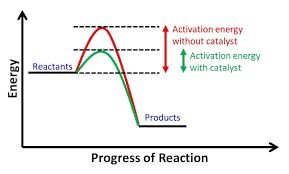
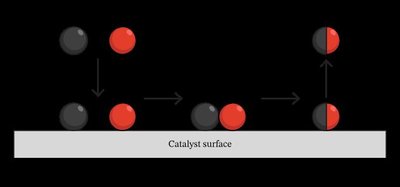
Catalysts
A catalyst is a substance that speeds up a reaction by providing an alternative pathway with lower activation energy. Catalysts are not consumed in the reaction and can work by offering a surface for the reaction to occur. In biological systems, catalysts are called enzymes.
Effect: Lowers activation energy, increasing the number of particles able to react.
Not consumed: Catalysts are not reactants or products.
Enzymes: Biological catalysts that speed up reactions in living organisms.
Summary Table: Factors Affecting Reaction Rate
Factor | Effect on Reaction Rate | Mechanism |
|---|---|---|
Temperature | Increases rate | More frequent and energetic collisions; more particles exceed activation energy |
Surface Area | Increases rate | More exposed particles; higher collision frequency |
Concentration | Increases rate | Higher particle density; more collisions per second |
Catalyst | Increases rate | Lowers activation energy; more particles can react |
Key Equations
Rate of Reaction:
Arrhenius Equation:
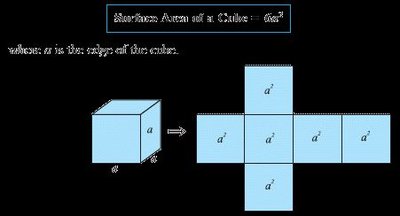
Surface Area of a Cube:
Conclusion
Controlling reaction rates is essential in chemistry. By understanding collision theory and the factors affecting reaction rates, chemists can manipulate conditions to achieve desired outcomes efficiently and safely.
