 Back
BackChemical Reactions and Equations: Evidence, Types, and Balancing
Study Guide - Smart Notes

Chemical Reactions: Evidence and Types
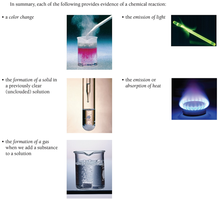
Evidence of a Chemical Reaction
Chemical reactions involve the transformation of one or more substances into different substances. Recognizing a chemical reaction is fundamental in chemistry. Several observable changes can indicate that a chemical reaction has occurred:
Color Change: A new color appears, indicating the formation of a new substance.
Formation of a Solid (Precipitate): A solid forms in a previously clear solution.
Formation of a Gas: Bubbles or gas are produced when substances are mixed.
Emission of Light: Light is produced during the reaction.
Emission or Absorption of Heat: The reaction releases or absorbs heat, causing a temperature change.
However, not all observable changes are evidence of a chemical reaction. For example, boiling water produces bubbles (gas), but no new substance is formed—this is a physical change, not a chemical one.


Chemical Equations and Balancing
Representing Chemical Reactions
Chemical reactions are represented by chemical equations. The substances present before the reaction are called reactants, and those formed are products. The state of each substance is indicated in parentheses: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous (dissolved in water).
Balancing Chemical Equations
Balancing chemical equations ensures the law of conservation of mass is obeyed—atoms are neither created nor destroyed. The number of each type of atom must be the same on both sides of the equation. To balance an equation, adjust the coefficients (numbers in front of formulas), not the subscripts (numbers within formulas).
Write the correct formulas for all reactants and products.
Balance elements that appear in only one reactant and one product first.
Balance free elements last.
Check your work by counting atoms of each element on both sides.
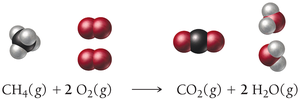
Example: Combustion of Methane
The reaction of methane with oxygen forms carbon dioxide and water:
Unbalanced equation:
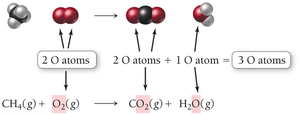
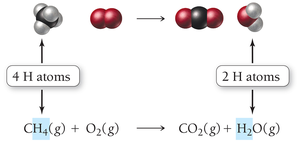
Balanced equation:
Types of Chemical Reactions
Precipitation Reactions
Precipitation reactions occur when two aqueous solutions combine to form an insoluble solid, called a precipitate. The formation of a solid in a previously clear solution is evidence of such a reaction.

Gas Evolution Reactions
Gas evolution reactions produce a gas as one of the products. For example, mixing baking soda and vinegar produces carbon dioxide gas, causing bubbling and fizzing.

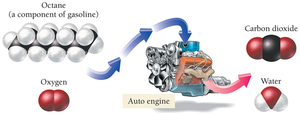
Combustion Reactions
Combustion reactions involve a substance reacting with oxygen to produce energy (heat and light), carbon dioxide, and water. These are a type of oxidation–reduction (redox) reaction.
Other Types of Reactions
Acid–Base Reactions: An acid reacts with a base to form water and a salt.
Oxidation–Reduction (Redox) Reactions: Involve the transfer of electrons between substances.
Synthesis Reactions: Two or more simple substances combine to form a more complex substance.
Decomposition Reactions: A complex substance breaks down into simpler substances.
Single-Displacement Reactions: One element replaces another in a compound.
Double-Displacement Reactions: Elements or groups in two compounds exchange places.
Chemical Reactions in Everyday Life
Soap and Detergent Reactions
Soap reacts with ions in hard water (calcium and magnesium) to form an insoluble residue (soap scum), while detergents contain substances that remove these ions, preventing residue formation.

Summary Table: States of Matter in Chemical Equations
Abbreviation | Meaning |
|---|---|
(s) | Solid |
(l) | Liquid |
(g) | Gas |
(aq) | Aqueous (dissolved in water) |
Key Points for Exam Preparation
Identify evidence of chemical reactions (color change, precipitate, gas, light, heat).
Write and balance chemical equations using correct formulas and coefficients.
Classify reactions: precipitation, acid–base, gas evolution, redox, combustion, synthesis, decomposition, single- and double-displacement.
Understand the importance of chemical equations in representing matter conservation.
