 Back
BackChemical Reactions and Stoichiometry: Study Guide for Introductory Chemistry
Study Guide - Smart Notes

Chapter 7: Chemical Reactions
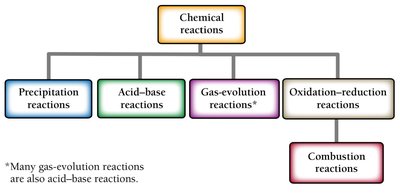
Chemical Reactions: Types and Definitions
Chemical reactions involve the transformation of one or more substances into different substances. Understanding the various types of reactions is fundamental in chemistry.
Chemical reaction: The process in which substances (reactants) are converted into new substances (products).
Gas-evolution reaction: Occurs in liquids and forms gases as products.
Combustion reaction: Involves oxygen, emits heat, and forms one or more oxygen-containing compounds.
Precipitation reaction: Forms solid substances (precipitates) in water.
Balancing Chemical Equations
Balancing equations ensures the conservation of mass by making sure the number of atoms of each element is equal on both sides of the equation.
States of matter are indicated in parentheses: (g) for gas, (l) for liquid, (s) for solid, (aq) for aqueous solution.
Balance equations by adjusting coefficients, not subscripts.
Each type of atom must be balanced.
Example:
Aqueous Solutions and Electrolytes
An aqueous solution is a homogeneous mixture of a substance with water. The ability of a substance to conduct electricity in solution depends on its ionization.
Electrolyte: Dissolves in water to form a solution that conducts electricity.
Non-electrolyte: Dissolves in water but does not conduct electricity.
Strong electrolyte: Completely dissociates into ions.
Weak electrolyte: Partially ionizes into ions.
Acid: Molecular substance that ionizes in water to form H+ ions.
Strong acid: Completely ionizes in water.
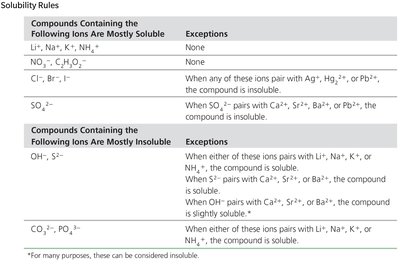
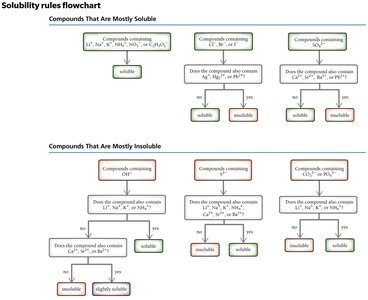
Solubility: A compound is soluble if it dissolves in a liquid; insoluble if it does not.
Soluble ionic compounds and strong acids produce strong electrolyte solutions.
Solubility Rules
Solubility rules help predict whether a compound will dissolve in water or form a precipitate.
Precipitation Reactions
Precipitation reactions occur when two aqueous solutions are mixed and an insoluble compound forms as a solid precipitate.
Only insoluble compounds form precipitates.
Write equations by pairing cations and anions.
Types of Chemical Equations
Chemical reactions can be represented in different ways to show the actual species involved.
Molecular equation: Shows complete neutral formulas for every compound.
Complete ionic equation: Shows reactants and products as they are present in solution (aqueous ionic compounds are separated into ions).
Net ionic equation: Shows only the species that participate in the reaction (removes spectator ions).
Spectator ions: Ions that do not participate in the reaction and appear unchanged on both sides.
Acid–Base and Gas-Evolution Reactions
Acid–base reactions form water when an acid and a base are mixed. Gas-evolution reactions produce a gas as a product.
Acid–base reaction:
Gas-evolution reaction:
Oxidation–Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between substances. Combustion reactions are a type of redox reaction.
Oxidation: Loss of electrons.
Reduction: Gain of electrons.
Mnemonics: OIL RIG (Oxidation Is Loss; Reduction Is Gain), LEO GER (Lose Electrons Oxidation; Gain Electrons Reduction).
Combustion reactions: Reaction with oxygen to form oxygen-containing compounds, often including water; exothermic (emit heat).
Classification of Chemical Reactions
Chemical reactions can be classified based on the changes occurring in the reactants and products.
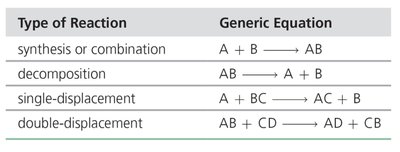
Synthesis (Combination) reaction: Simple substances combine to form more complex substances. Generic equation:
Decomposition reaction: A complex substance breaks down into simpler substances. Generic equation:
Single-displacement reaction: One element displaces another in a compound. Generic equation:
Double-displacement reaction: Two elements or groups in different compounds exchange places. Generic equation:
Chapter 8: Quantities in Chemical Reactions (Stoichiometry)
Reaction Stoichiometry
Stoichiometry is the numerical relationship between chemical quantities in a balanced chemical equation. It allows chemists to predict the amounts of reactants needed or products formed in a reaction.
Based on balanced equations, stoichiometry relates moles of reactants and products.
Used to calculate how much of a reactant is required to react with another, or how much product will form.
Example: In the reaction , 2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water.
Additional info: Stoichiometry is essential for laboratory calculations, industrial chemical production, and understanding reaction yields.
