 Back
BackChemical Reactions, Equations, and Aqueous Solutions: Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Chapter 7: Chemical Reactions and Equations
Evidence of a Chemical Reaction
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products). Recognizing a chemical reaction is fundamental in chemistry, and several observable changes can indicate that a reaction has occurred.
Color Change: A visible shift in color often signals a chemical transformation.
Formation of a Solid (Precipitate): The appearance of a solid in a previously clear solution is evidence of a reaction.
Formation of a Gas: Bubbling or fizzing when substances are mixed can indicate gas evolution.
Emission of Light: Some reactions produce light as a product.
Emission or Absorption of Heat: Temperature changes may accompany chemical changes.
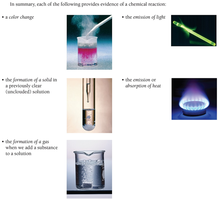
It is important to note that not all observable changes are chemical reactions. For example, boiling water produces bubbles, but this is a physical change, not a chemical one.


Types of Chemical Reactions
Chemical reactions can be classified based on the changes that occur and the products formed.
Gas Evolution Reactions: Reactions in which a gas is formed, such as the classic volcano experiment with baking soda and vinegar.
Precipitation Reactions: Reactions that produce a solid (precipitate) when two solutions are mixed.
Combustion Reactions: Reactions where a substance reacts with oxygen to produce heat, carbon dioxide, and water.
Acid–Base Reactions: Reactions between acids and bases that typically produce water and a salt.
Oxidation–Reduction (Redox) Reactions: Reactions involving the transfer of electrons between substances.

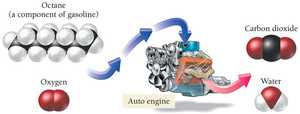


Chemical Equations
Chemical equations are symbolic representations of chemical reactions. They show the reactants on the left and the products on the right, separated by an arrow. States of matter are indicated in parentheses: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous (dissolved in water).
Reactants: Substances present before the reaction.
Products: Substances formed as a result of the reaction.
States: Indicated as (s), (l), (g), or (aq).
Balancing Chemical Equations
Balancing ensures the law of conservation of mass is obeyed: the number of atoms of each element must be the same on both sides of the equation.
Insert coefficients (whole numbers) in front of formulas to balance atoms.
Never change the subscripts in chemical formulas.
Balance elements that appear in only one compound first; balance free elements last.
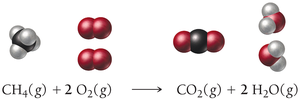
Example: Combustion of methane
Unbalanced equation:
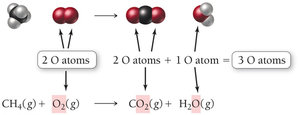
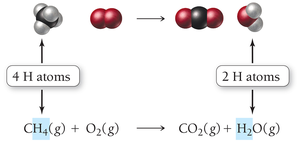
Balanced equation:
Precipitation Reactions and Solubility
Precipitation reactions occur when two aqueous solutions are mixed and an insoluble solid forms. The solubility of ionic compounds in water is determined by empirical rules.
Soluble compounds: Dissolve in water to form ions (strong electrolytes).
Insoluble compounds: Do not dissolve; remain as solids.

Writing Chemical Equations for Reactions in Solution
Molecular Equation: Shows all reactants and products as neutral compounds.
Complete Ionic Equation: Shows all strong electrolytes as ions.
Net Ionic Equation: Shows only the species that actually participate in the reaction (spectator ions are omitted).
Classifying Chemical Reactions by Atom Rearrangement
Synthesis Reaction: Two or more simple substances combine to form a more complex substance.
Decomposition Reaction: A complex substance breaks down into simpler substances.
Single-Displacement Reaction: One element replaces another in a compound.
Double-Displacement Reaction: Elements or groups in two compounds exchange places.
Summary Table: Types of Chemical Reactions
Type | Description | General Form |
|---|---|---|
Synthesis | Two substances combine | |
Decomposition | One substance breaks down | |
Single-Displacement | Element replaces another | |
Double-Displacement | Exchange of groups | |
Combustion | Reaction with oxygen |
Key Learning Objectives
Identify evidence of chemical reactions.
Write and balance chemical equations.
Determine solubility and predict precipitation reactions.
Write molecular, complete ionic, and net ionic equations.
Classify chemical reactions by type.
