 Back
BackChemical Reactions: Evidence, Types, and Equations
Study Guide - Smart Notes

Chemical Reactions
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products) through the breaking and forming of chemical bonds. These reactions are fundamental to both natural and industrial processes, such as the combustion of fuel in engines and the metabolism of food in living organisms.
Evidence of a Chemical Reaction
Observable Signs of Chemical Change
Several observable changes can indicate that a chemical reaction has occurred. However, only chemical analysis can conclusively prove that new substances have formed.
Color changes: A new color appears or an existing color fades.
Formation of a precipitate: A solid forms in a previously clear solution.
Gas formation: Bubbles or effervescence indicate the production of a gas.
Light emission: Light is produced, such as in combustion or glow sticks.
Heat emission or absorption: The reaction releases or absorbs heat.

Physical Changes vs. Chemical Changes
Not all observable changes are evidence of a chemical reaction. For example, boiling water produces bubbles, but this is a physical change—water molecules remain unchanged.


Atomic and Molecular Level Changes
At the atomic and molecular level, a chemical reaction involves the rearrangement of atoms to form new substances. Only chemical analysis can confirm the transformation of reactants into products.
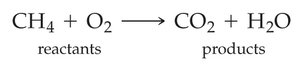
The Chemical Equation
Structure of a Chemical Equation
Chemical equations represent chemical reactions using chemical formulas. The substances on the left are reactants, and those on the right are products. States of matter are indicated in parentheses: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution.
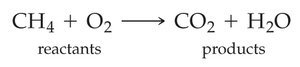
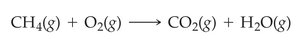
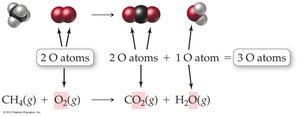
Balancing Chemical Equations
Balancing ensures the same number of each type of atom on both sides of the equation, reflecting the law of conservation of mass. Only coefficients (not subscripts) are changed to balance equations.
Write the unbalanced equation.
Balance the equation by adjusting coefficients.
Never change subscripts in chemical formulas.
Indicate the states of matter for all reactants and products.
Aqueous Solutions and Solubility
Solubility and Electrolytes
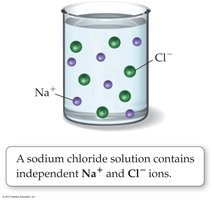
A compound is soluble if it dissolves in a liquid, and insoluble if it does not. An aqueous solution is a homogeneous mixture of a substance with water. Ionic compounds that dissolve in water usually dissociate into ions, forming strong electrolytes that conduct electricity.

Examples of Dissolution
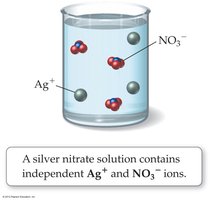
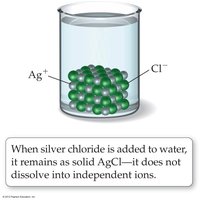
When AgNO3 dissolves in water, it dissociates into Ag+ and NO3− ions. Not all ionic compounds are soluble; for example, AgCl does not dissolve in water and is not an electrolyte.
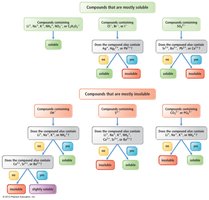
Solubility Rules
Solubility rules help predict whether an ionic compound will dissolve in water. These rules are summarized in tables and flowcharts for quick reference.
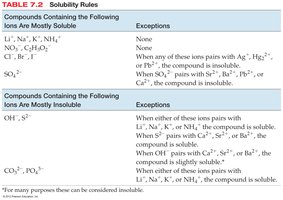
Compounds Containing the Following Ions | Are Mostly Soluble | Exceptions |
|---|---|---|
Li+, Na+, K+, NH4+ | Yes | None |
NO3−, C2H3O2−, ClO4− | Yes | None |
Cl−, Br−, I− | Yes | With Ag+, Hg22+, Pb2+ |
SO42− | Yes | With Sr2+, Ba2+, Pb2+, Ca2+ |
Compounds Containing the Following Ions | Are Mostly Insoluble | Exceptions |
|---|---|---|
OH−, S2− | Yes | With Li+, Na+, K+, NH4+; S2− with Ca2+, Sr2+, Ba2+; OH− with Ca2+, Sr2+, Ba2+ (slightly soluble) |
CO32−, PO43− | Yes | With Li+, Na+, K+, NH4+ |
Precipitation Reactions
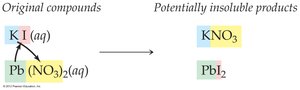
Formation of a Precipitate
Precipitation reactions occur when two aqueous solutions combine to form an insoluble solid, called a precipitate. The key to predicting these reactions is to use solubility rules to determine if any product is insoluble.
Mixing solutions of soluble compounds may result in an insoluble product.
If all products are soluble, no reaction occurs.

Writing Equations for Precipitation Reactions
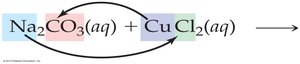
To write these equations:
Write the formulas of the reactants and products, switching cations and anions.
Use solubility rules to identify the precipitate.
Balance the equation.
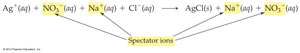
Chemical Equations in Solution
Molecular, Complete Ionic, and Net Ionic Equations
There are three main ways to represent reactions in solution:
Molecular equation: Shows complete, neutral formulas for all compounds.
Complete ionic equation: Shows all strong electrolytes as ions.
Net ionic equation: Shows only the species that actually participate in the reaction (spectator ions are omitted).
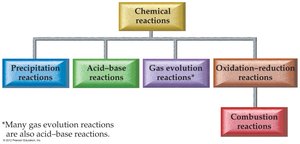
Classifying Chemical Reactions
Main Types of Chemical Reactions
Chemical reactions can be classified into several main types based on the changes that occur:
Precipitation reactions: Formation of an insoluble solid.
Acid–base reactions: Formation of water from an acid and a base.
Gas evolution reactions: Formation of a gas.
Oxidation–reduction (redox) reactions: Transfer of electrons between substances.
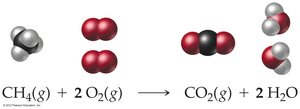
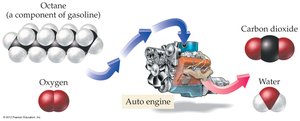
Combustion reactions: A type of redox reaction involving O2 and producing heat, CO2, and H2O.
Acid–Base Reactions
Acid–base (neutralization) reactions involve the reaction of an acid with a base to form water and a salt. The net ionic equation for many acid–base reactions is:
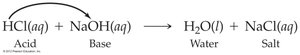
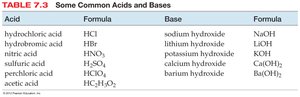
Acid | Formula | Base | Formula |
|---|---|---|---|
hydrochloric acid | HCl | sodium hydroxide | NaOH |
hydrobromic acid | HBr | lithium hydroxide | LiOH |
nitric acid | HNO3 | potassium hydroxide | KOH |
sulfuric acid | H2SO4 | calcium hydroxide | Ca(OH)2 |
perchloric acid | HClO4 | barium hydroxide | Ba(OH)2 |
acetic acid | HC2H3O2 |
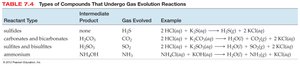
Gas Evolution Reactions
Gas evolution reactions produce a gas as a product. The type of gas formed depends on the reactants. Common gases evolved include H2S, CO2, SO2, and NH3.
Reactant Type | Intermediate Product | Gas Evolved | Example |
|---|---|---|---|
sulfides | none | H2S | 2KI(aq) + H2SO4(aq) → H2S(g) + 2KNO3(aq) |
carbonates and bicarbonates | H2CO3 | CO2 | NaHCO3(aq) + HCl(aq) → CO2(g) + H2O(l) + NaCl(aq) |
sulfites and bisulfites | H2SO3 | SO2 | NaHSO3(aq) + HCl(aq) → SO2(g) + H2O(l) + NaCl(aq) |
ammonium | NH4OH | NH3 | NH4Cl(aq) + NaOH(aq) → NH3(g) + H2O(l) + NaCl(aq) |
Oxidation–Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between substances. Oxidation is the loss of electrons, while reduction is the gain of electrons. These processes always occur together.
Combustion reactions are a type of redox reaction, characterized by the reaction of a substance with O2 to form oxygen-containing compounds and release heat.
General Types of Reactions by Atom Rearrangement
Type of Reaction | Generic Equation |
|---|---|
Synthesis or combination | A + B → AB |
Decomposition | AB → A + B |
Displacement | A + BC → AC + B |
Double-displacement | AB + CD → AD + CB |

