 Back
BackComprehensive Study Guide: Types of Chemical Reactions and Acid-Base Chemistry
Study Guide - Smart Notes

Examining Chemical Reactions
Introduction to Chemical Reactions
Chemical reactions are processes in which substances (reactants) are transformed into new substances (products) with different properties. Understanding the main types of chemical reactions is fundamental in introductory chemistry, as it provides the basis for predicting products and balancing equations.
Types of Chemical Reactions
Synthesis (Combination) Reactions
In a synthesis reaction, two or more simple substances combine to form a more complex compound. This is a fundamental process for building new materials in chemistry.
General Formula:
Key Feature: Two reactants form one product.
Example: The formation of rust (iron(III) oxide) from iron and oxygen:

Decomposition Reactions
Decomposition reactions involve a single compound breaking down into two or more simpler substances. These reactions often require energy input, such as heat, light, or electricity.
General Formula:
Key Feature: One reactant splits into two or more products.
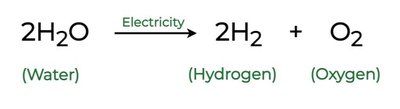
Example: Electrolysis of water:

Combustion Reactions
Combustion reactions occur when a substance reacts rapidly with oxygen, releasing energy in the form of light and heat. Hydrocarbon combustion always produces carbon dioxide and water.
General Formula:
Key Feature: Exothermic reaction producing heat and light.
Example: Burning methane gas:

Single Displacement (Replacement) Reactions
In single displacement reactions, an uncombined element replaces another element in a compound. Metals replace metals, and non-metals replace non-metals.
General Formula:
Key Feature: One element swaps places with another in a compound.
Example: Iron nails placed in copper(II) sulfate solution:

Double Displacement (Metathesis) Reactions
Double displacement reactions involve the exchange of ions between two compounds, resulting in the formation of two new compounds. This often leads to the formation of a precipitate, gas, or water.
General Formula:
Key Feature: Cations and anions switch partners.
Example: Mixing solutions of lead(II) nitrate and potassium iodide forms a yellow precipitate of lead(II) iodide.


Neutralization (Acid-Base) Reactions
Neutralization is a specific type of double displacement reaction where an acid reacts with a base to produce water and a salt.
General Formula:
Key Feature: Formation of water and a salt.
Example: Hydrochloric acid reacts with magnesium hydroxide:
Precipitation Reactions
Precipitation reactions occur when two aqueous solutions react to form an insoluble solid, called a precipitate. The formation of a solid indicates the reaction has occurred.
Key Feature: Formation of an insoluble solid from two solutions.
Example: Mixing solutions of silver nitrate and sodium chloride forms a white precipitate of silver chloride:
Summary Table: Types of Chemical Reactions
Reaction Type | General Formula | Key Feature |
|---|---|---|
Synthesis | A + B → AB | Two become one |
Decomposition | AB → A + B | One splits into two |
Combustion | Fuel + O₂ → CO₂ + H₂O | Fire, heat, CO₂ |
Single Displacement | A + BC → AC + B | The "Swap" |
Double Displacement | AB + CD → AD + CB | Switching Partners |
Acids, Bases, and Their Reactions
Common Laboratory Acids and Their Salts
Acids react with bases and metals to form salts. The name of the salt depends on the acid used:
Name of Acid | Molecular Formula | Name of Salt Formed |
|---|---|---|
Hydrochloric acid | HCl | Chloride |
Sulphuric acid | H₂SO₄ | Sulphate |
Nitric acid | HNO₃ | Nitrate |
Types of Bases
Metal Oxides
Metal Hydroxides
Metal Carbonates
Products of Neutralization Reactions
When acids react with bases, the products are always a salt and water. If the base is a carbonate, carbon dioxide is also produced.

Acid Reactions with Metals
More reactive metals in the periodic table react with acids to produce a salt and hydrogen gas.

Naming Salts from Acid Reactions
The name of the salt formed in an acid reaction is determined by the metal and the acid used. The metal gives the first part of the name, and the acid gives the ending (chloride, sulphate, nitrate).

Examples of Acid Reactions
Reactants | Products |
|---|---|
Hydrochloric Acid + Iron Oxide | Iron Chloride + Water |
Sulphuric Acid + Sodium Hydroxide | Sodium Sulphate + Water |
Nitric Acid + Calcium Carbonate | Calcium Nitrate + Water + Carbon dioxide |
Hydrochloric Acid + Magnesium | Magnesium Chloride + Hydrogen |

Practice: Writing Word and Balanced Equations
Students should be able to write word equations and balanced chemical equations for reactions between acids and bases, metals, and carbonates. Include state symbols where appropriate.


Additional info: Understanding the types of chemical reactions and the rules for naming salts is essential for predicting the outcomes of chemical processes and for laboratory work in introductory chemistry.
