 Back
BackElectrons in Atoms and the Periodic Table: Study Guide
Study Guide - Smart Notes

Electrons in Atoms and the Periodic Table
Wavelength and Frequency
Light energy travels through space as electromagnetic radiation, which can be described as both particles and waves. The speed of light in a vacuum is a fundamental constant. The structure of a wave includes the crest (top) and trough (bottom). Wavelength (λ, Greek lambda) is the distance from one crest or trough to the next, measured in meters or nanometers. Frequency (ν, Greek nu) is the number of waves passing a point per second, measured in Hertz (Hz).
Relationship: At a fixed speed, frequency is inversely proportional to wavelength and directly proportional to energy.
High frequencies: Short wavelengths, high energies.
Low frequencies: Long wavelengths, low energies.
Formula: (where is the speed of light, is wavelength, is frequency)

Example: The wave with the shortest wavelength (e.g., 325 nm) has the highest frequency.
Electromagnetic Spectrum
The Electromagnetic Spectrum is a continuum of electromagnetic radiation, including all wavelengths and frequencies. Electromagnetic radiation is the flow of energy at the speed of light through space as electric and magnetic fields. Max Planck and Albert Einstein described this radiation as being made of "packets" or "particles" called quanta (photons).
As we move from radio waves to gamma rays, wavelengths decrease and frequencies increase.
The Visible Light Spectrum is the small portion of the spectrum visible to the human eye.
Energy: Higher frequency radiation (e.g., gamma rays) has higher energy.

Example: X-rays and gamma rays have the greatest energy per photon.
Bohr Model of the Atom
The Bohr Model describes electrons traveling around the nucleus in circular orbits called shells. Each shell (n) represents a grouping of electrons with a specific potential energy. Electrons can move between shells by absorbing or emitting energy.
Absorption: Electron moves from a lower-numbered shell to a higher-numbered shell.
Emission: Electron moves from a higher-numbered shell to a lower-numbered shell.
As the distance between shells increases, the energy involved increases.
Formula for energy transitions:
Example: The electron furthest from the nucleus has the highest n value.
Emission Spectrum
An Emission Spectrum is a series of lines formed when emitted light is focused by a slit and passed through a prism. Each line corresponds to a specific energy transition in the atom.
Slit: Spreads closely packed wavelengths.
Prism: Separates light into discrete lines.
Lines are due to electrons moving from higher to lower energy states.

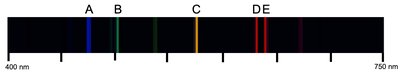
Example: The line with the shortest wavelength (highest frequency) has the highest energy.
Electronic Structure: Shells, Subshells, and Orbitals
The modern description of atomic structure includes shells, subshells, and orbitals:
Shell: Main energy level (n).
Subshell: Subdivision of a shell, designated by letters (s, p, d, f).
Orbital: Region within a subshell where electrons are likely to be found.
Each shell can be divided into subshells, and each subshell contains a set of orbitals.
Example: The third shell (n = 3) contains three subshells: 3s, 3p, and 3d.
Orbital Shapes and Sets
Each subshell has a characteristic shape:
s: Spherical
p: Dumbbell-shaped
d: Cloverleaf-shaped
f: Complex shapes (beyond scope)
Each set of orbitals has a specific number: s (1), p (3), d (5), f (7)
Example: The 4th energy level and p sublevel contains three 4p orbitals.
Electron Spin and the Pauli Exclusion Principle
Each orbital can hold a maximum of two electrons with opposite spins, according to the Pauli Exclusion Principle. Electron spin is either +1/2 (up) or –1/2 (down).
No two electrons in the same orbital can have the same spin.
Electrons fill orbitals singly before pairing (Hund's Rule).
Example: In a set of 5d orbitals, electrons are distributed according to their spin and energy levels.
Number of Electrons in Shells
Each shell can hold a maximum number of electrons, given by:
Formula: (where n is the shell number)
Example: The 6th shell can hold electrons.
Electron Configuration
Electron configurations describe the arrangement of electrons in an atom's orbitals. The Auf Bau Principle states that electrons fill the lowest energy orbitals first.
Order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, etc.
Hund's Rule: Orbitals of the same energy are singly occupied before any is doubly occupied.
Condensed configuration uses the noble gas preceding the element in brackets.
Example: Fluorine (Z = 9): 1s2 2s2 2p5
Unpaired vs. Paired Electrons
Orbitals can contain unpaired or paired electrons:
Unpaired: One electron with its own spin.
Paired: Two electrons with opposite spins.
Example: Vanadium has three unpaired electrons in its ground state.
Ions and the Octet Rule
Main group elements tend to achieve eight valence electrons (a full outer shell) by gaining or losing electrons:
Metals: Lose electrons to resemble the preceding noble gas.
Non-metals: Gain electrons to resemble the following noble gas.
Electron configurations for ions are adjusted by adding or removing electrons from the highest shell.
Example: Magnesium (Z = 12) loses two electrons to achieve a filled outer shell.
Valence Electrons of Elements
Valence electrons are the outer shell electrons involved in chemical bonding. The number of valence electrons for main group elements equals their group number.
Inner core electrons: All other electrons not in the outer shell.
Formula: Atomic number (Z) = Valence electrons + Inner core electrons
Example: Carbon has four valence electrons.
Periodic Trends: Metallic Character
Periodic trends describe patterns in element properties:
Metallic character: How easily an element loses an electron.
Metals lose electrons; non-metals gain electrons.
Metallic character decreases from left to right across a period and increases down a group.
Example: Rubidium has greater metallic character than oxygen.
Periodic Trends: Atomic Radius
Atomic radius is the distance from the nucleus to the outer electron shell. It increases down a group and decreases across a period.
More electrons and shells down a group increase radius.
More electrons in the same shell across a period increase attraction to the nucleus, decreasing radius.
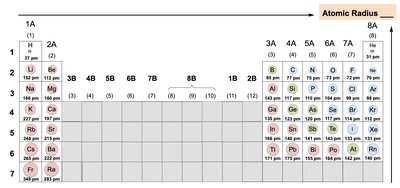
Example: Rubidium has a larger atomic radius than calcium.
Periodic Trends: Ionization Energy
Ionization energy is the energy required to remove an electron from a gaseous atom. It increases across a period and decreases down a group.
Low IE: Electron easily lost.
High IE: Electron not easily lost.
Noble gases have the highest IE due to stable configurations.
Example: Potassium has a lower ionization energy than fluorine.
Periodic Trends: Electron Affinity
Electron affinity is the energy released when an electron is added to a gaseous atom. It increases across a period and decreases down a group.
High EA: Electron easily accepted.
Low EA: Electron not easily accepted.
Exceptions: Noble gases have low EA due to stable configurations.
Example: Fluorine releases the most energy when accepting an electron among the halogens.
*Additional info: Some explanations and formulas were expanded for clarity and completeness based on standard introductory chemistry textbooks.*
