 Back
BackIntro to Chemistry: Solutions, Solubility, and Electrolytes Study Guide
Study Guide - Smart Notes

Q1. What happens when sugar is dissolved in water? How is it different when NaCl or HCl dissolve in water?
Background
Topic: Electrolytes vs. Nonelectrolytes; Dissolving Process
This question tests your understanding of how different types of substances (molecular vs. ionic) behave when dissolved in water, and how this affects their ability to conduct electricity.
Key Terms and Concepts:
Electrolyte: A substance that produces ions in solution and conducts electricity.
Nonelectrolyte: A substance that does not produce ions in solution and does not conduct electricity.
Dissociation: The process by which ionic compounds separate into ions in solution.
Ionization: The process by which a molecular compound forms ions in solution (e.g., HCl in water).
Step-by-Step Guidance
Consider what happens at the molecular level when sugar (a covalent compound) dissolves in water. Does it break into ions or stay as molecules?
Think about the electrical conductivity of a sugar solution. Would a light bulb connected to the solution light up?
Now, consider what happens when NaCl (an ionic compound) dissolves in water. Does it dissociate into ions?
Think about the electrical conductivity of a NaCl solution. Would a light bulb connected to the solution light up?
Finally, consider HCl (a molecular compound that ionizes in water). What ions are produced, and how does this affect conductivity?
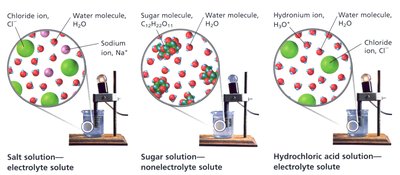
Try solving on your own before revealing the answer!
Final Answer:
When sugar dissolves in water, it stays as whole molecules and does not conduct electricity (nonelectrolyte). When NaCl or HCl dissolve, they produce ions (Na+, Cl-, H3O+, Cl-), allowing the solution to conduct electricity (electrolytes).
NaCl dissociates into ions, while HCl ionizes to form ions in water.
Q2. What happens if 46.4 g NaCH3COO is added to 100. g of 20°C water? What happens if you add 46.5 g? Would stirring help dissolve more?
Background
Topic: Solubility, Saturated and Unsaturated Solutions
This question tests your understanding of the concepts of solubility, saturated solutions, and the effect of stirring on dissolving solids.
Key Terms and Concepts:
Solubility: The maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature.
Saturated Solution: Contains the maximum amount of dissolved solute at a given temperature.
Unsaturated Solution: Can dissolve more solute at the same temperature.
Stirring: Increases the rate of dissolution but does not increase the maximum solubility.
Step-by-Step Guidance
Check the solubility of NaCH3COO in 100 g of water at 20°C (from the graph or data provided).
Compare the amount added (46.4 g) to the solubility limit. Is the solution saturated or unsaturated?
Consider what happens if you add slightly more (46.5 g). Will all of it dissolve, or will some remain undissolved?
Think about the effect of stirring on the amount that can dissolve versus the rate at which it dissolves.

Try solving on your own before revealing the answer!
Final Answer:
46.4 g is the solubility limit at 20°C, so all of it will dissolve, making a saturated solution. If you add 46.5 g, a tiny amount (0.1 g) will remain undissolved. Stirring helps dissolve the solute faster but does not increase the maximum amount that can dissolve.
Q3. How many grams of KI will dissolve in 100 ml of water at 45°C? How many grams of NaCl will dissolve in 50 grams of water at 75°C? What substance has a solubility of 90 g/100 cm³ of water at a temperature of 25°C?
Background
Topic: Reading Solubility Curves
This question tests your ability to interpret solubility graphs and apply proportional reasoning to determine how much solute will dissolve under different conditions.
Key Terms and Concepts:
Solubility Curve: A graph showing how much solute can dissolve in a given amount of solvent at various temperatures.
Proportional Reasoning: Adjusting solubility values for different amounts of solvent.
Step-by-Step Guidance
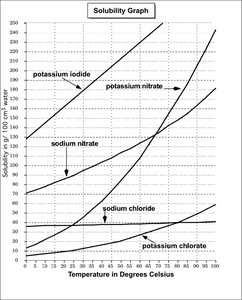
Locate the solubility of KI at 45°C on the solubility graph (in g/100 ml water).
For NaCl at 75°C, find the solubility value on the graph (in g/100 g water), then calculate how much will dissolve in 50 g of water using a proportion.
For the third part, look for the substance whose solubility is 90 g/100 cm³ at 25°C on the graph.
Try solving on your own before revealing the answer!
Final Answer:
1) The solubility of KI at 45°C is approximately 180 g/100 ml water. 2) The solubility of NaCl at 75°C is about 40 g/100 g water, so in 50 g water, about 20 g will dissolve. 3) Potassium nitrate (KNO3) has a solubility of 90 g/100 cm³ at 25°C.
