 Back
BackIntro to Chemistry Study Guide: Periodic Table, Compounds, and Reactions
Study Guide - Smart Notes

Q1. Based on the general trends in the periodic table, circle the element in each of the following pairs that has the most metallic character:
Background
Topic: Periodic Table Trends
This question tests your understanding of how metallic character changes across periods and down groups in the periodic table.
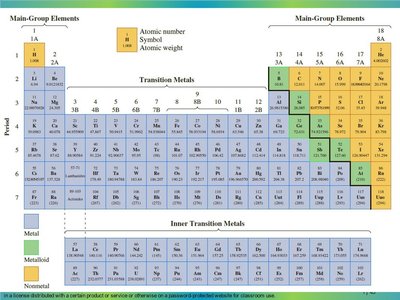
Key Terms:
Metallic character: The tendency of an element to lose electrons and form positive ions (cations).
Periodic trends: Metallic character increases down a group and decreases across a period (left to right).
Step-by-Step Guidance
Identify the position of each element in the pair on the periodic table.
Recall that metallic character increases as you move down a group and decreases as you move from left to right across a period.
Compare the elements in each pair based on their group and period placement.
Determine which element is further down or further to the left, as these are more metallic.
Try solving on your own before revealing the answer!
Q2. State the number of valence electrons for each of the following elements.
Background
Topic: Electron Configuration and Valence Electrons
This question tests your ability to determine the number of valence electrons for main-group elements using the periodic table.

Key Terms:
Valence electrons: Electrons in the outermost shell of an atom, important for chemical bonding.
Group number: For main-group elements, the group number often indicates the number of valence electrons.
Step-by-Step Guidance
Locate the element on the periodic table.
Identify its group number (for groups 1A–8A).
For main-group elements, the group number corresponds to the number of valence electrons (e.g., Group 1A = 1 valence electron).
Write down the number of valence electrons for each element.
Try solving on your own before revealing the answer!
Q3. According to general trends in the periodic table, circle the element in each of the following pairs that has the larger atomic radius.
Background
Topic: Atomic Radius Trends
This question tests your understanding of how atomic radius changes across periods and down groups in the periodic table.

Key Terms:
Atomic radius: The distance from the nucleus to the outermost electron shell.
Periodic trends: Atomic radius increases down a group and decreases across a period (left to right).
Step-by-Step Guidance
Identify the position of each element in the pair on the periodic table.
Recall that atomic radius increases as you move down a group and decreases as you move from left to right across a period.
Compare the elements in each pair based on their group and period placement.
Determine which element is further down or further to the left, as these have larger atomic radii.
Try solving on your own before revealing the answer!
Q4. Draw the electron dot formula for each of the following elements.
Background
Topic: Lewis Dot Structures
This question tests your ability to represent valence electrons as dots around the element symbol.
Key Terms:
Electron dot formula (Lewis dot structure): Shows the valence electrons as dots around the element symbol.
Valence electrons: Use the group number to determine how many dots to place.
Step-by-Step Guidance
Identify the element and its number of valence electrons.
Write the element symbol.
Place dots around the symbol to represent each valence electron (up to 8).
Arrange the dots in pairs as needed.
Try solving on your own before revealing the answer!
Q5. Refer to the periodic table and circle the element in each of the following pairs that has the higher ionization energy.
Background
Topic: Ionization Energy Trends
This question tests your understanding of how ionization energy changes across periods and down groups in the periodic table.

Key Terms:
Ionization energy: The energy required to remove an electron from an atom.
Periodic trends: Ionization energy increases across a period (left to right) and decreases down a group.
Step-by-Step Guidance
Identify the position of each element in the pair on the periodic table.
Recall that ionization energy increases as you move from left to right across a period and decreases as you move down a group.
Compare the elements in each pair based on their group and period placement.
Determine which element is further to the right or higher up, as these have higher ionization energies.
Try solving on your own before revealing the answer!
Q6. Refer to the periodic table and write the complete electron configuration for each of the following ions.
Background
Topic: Electron Configuration of Ions
This question tests your ability to write electron configurations for ions, considering the gain or loss of electrons.
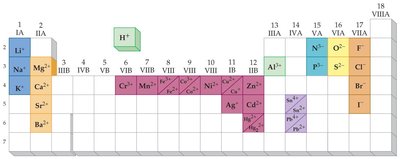
Key Terms and Formula:
Electron configuration: The arrangement of electrons in an atom or ion.
For cations: Remove electrons from the highest energy level.
For anions: Add electrons to the lowest available energy level.
Step-by-Step Guidance
Identify the neutral atom's electron configuration.
Determine if the ion is a cation (positive) or anion (negative).
For cations, remove electrons from the highest energy orbital; for anions, add electrons to the lowest available orbital.
Write the new electron configuration for the ion.
Try solving on your own before revealing the answer!
Q7. Provide the chemical formula for each of the following compounds:
Background
Topic: Chemical Formulas for Ionic and Covalent Compounds
This question tests your ability to write chemical formulas based on the names of compounds.

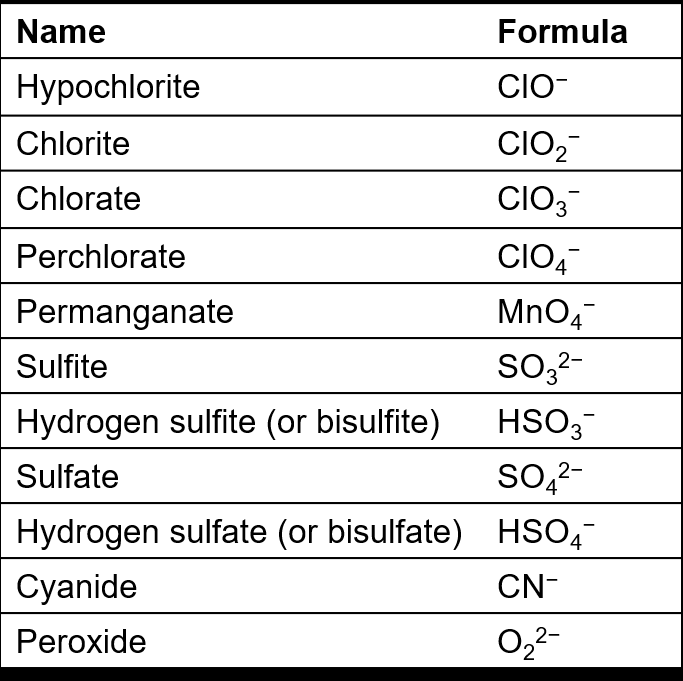
Key Terms:
Polyatomic ions: Ions made up of more than one atom, often used in compound formulas.
Formula writing: Combine cations and anions to balance charges.
Step-by-Step Guidance
Identify the cation and anion in the compound name.
Write the formula for each ion (refer to the tables for polyatomic ions).
Balance the charges so the total positive and negative charges equal zero.
Write the final chemical formula using subscripts as needed.
Try solving on your own before revealing the answer!
Q8. Provide the chemical formula for each of the acids:
Background
Topic: Chemical Formulas for Acids
This question tests your ability to write formulas for acids based on their names.


Key Terms:
Acid naming: Acids often contain hydrogen and a polyatomic ion.
Formula writing: Combine H+ with the anion to balance charges.
Step-by-Step Guidance
Identify the acid name and the corresponding anion.
Write the formula for the anion (refer to the tables for polyatomic ions).
Combine H+ ions with the anion to balance the charges.
Write the final chemical formula for the acid.
Try solving on your own before revealing the answer!
Q9. Supply a systematic name for each of the following compounds:
Background
Topic: Nomenclature of Compounds
This question tests your ability to name compounds systematically, including ionic and covalent compounds.
Key Terms:
Systematic name: The official IUPAC name for a compound.
Naming rules: Use the cation name first, then the anion name (with appropriate suffixes).
Step-by-Step Guidance
Identify the cation and anion in the formula.
Use the correct naming rules for ionic or covalent compounds.
Apply prefixes or suffixes as needed (e.g., -ide, -ate, -ite).
Write the systematic name for each compound.
Try solving on your own before revealing the answer!
Q10. Classify each of the following reactions:
Background
Topic: Types of Chemical Reactions
This question tests your ability to identify reaction types such as synthesis, decomposition, single replacement, double replacement, and combustion.
Key Terms:
Synthesis: Two or more substances combine to form one product.
Decomposition: One substance breaks down into two or more products.
Single replacement: One element replaces another in a compound.
Double replacement: Two compounds exchange ions.
Combustion: A substance reacts with oxygen to produce energy.
Step-by-Step Guidance
Examine the reactants and products in each equation.
Identify the pattern of the reaction (e.g., A + B → AB for synthesis).
Match the reaction to one of the main types.
Write the classification for each reaction.
Try solving on your own before revealing the answer!
Q11. Complete and balance each equation. If no reaction (no precipitation) occurs, write “NR”
Background
Topic: Balancing Chemical Equations and Precipitation Reactions
This question tests your ability to complete and balance chemical equations, and to predict whether a reaction occurs based on solubility rules.
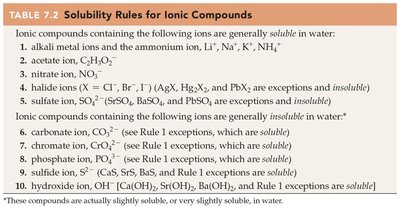
Key Terms and Formula:
Balancing equations: Ensure the same number of atoms of each element on both sides.
Solubility rules: Predict whether a product is soluble or forms a precipitate.
Step-by-Step Guidance
Write the reactants and predict the products based on reaction type.
Use solubility rules to determine if a precipitate forms (if not, write "NR").
Balance the equation by adjusting coefficients.
Check that all atoms and charges are balanced.
Try solving on your own before revealing the answer!
Q12. Predict whether or not a reaction occurs for each of the following:
Background
Topic: Predicting Chemical Reactions
This question tests your ability to use solubility rules and activity series to predict whether a reaction will occur.

Key Terms:
Solubility rules: Used to predict precipitation reactions.
Activity series: Used to predict single replacement reactions.
Step-by-Step Guidance
Identify the type of reaction (precipitation, single replacement, etc.).
Use solubility rules to check if a product is insoluble.
Use the activity series to check if a metal can replace another.
Predict whether a reaction occurs or not.
Try solving on your own before revealing the answer!
Q13. Predict which of the following metals reacts with water at room temperature.
Background
Topic: Reactivity of Metals
This question tests your ability to use the activity series to predict which metals react with water.
Key Terms:
Activity series: A list of metals ranked by their reactivity.
Active metals: Metals that react with water to release hydrogen gas.
Step-by-Step Guidance
Refer to the activity series provided.
Identify which metals are listed as reacting with water at room temperature.
Compare the metals in the question to the list.
Circle those that react with water.
Try solving on your own before revealing the answer!
Q14. State whether the following compounds are soluble or insoluble in water.
Background
Topic: Solubility Rules
This question tests your ability to use solubility rules to determine if a compound is soluble or insoluble in water.

Key Terms:
Solubility: The ability of a compound to dissolve in water.
Solubility rules: Guidelines for predicting solubility of ionic compounds.
Step-by-Step Guidance
Identify the ions present in the compound.
Refer to the solubility rules table to check if the compound is generally soluble or insoluble.
Look for any exceptions listed in the rules.
State whether the compound is soluble or insoluble.
Try solving on your own before revealing the answer!
Q15. Complete and balance each of the following neutralization reactions.
Background
Topic: Neutralization Reactions
This question tests your ability to write and balance equations for acid-base reactions.
Key Terms and Formula:
Neutralization: Reaction between an acid and a base to form water and a salt.
General equation:
Step-by-Step Guidance
Write the formulas for the acid and base.
Predict the products: water and an ionic salt.
Balance the equation by adjusting coefficients.
Check that all atoms and charges are balanced.
Try solving on your own before revealing the answer!
