 Back
BackIntroduction to Chemistry: Matter, Mixtures, Elements, and Transformations
Study Guide - Smart Notes

Chapter 1: What is Chemistry?
Science and Technology
Chemistry is often referred to as the central science because it bridges physics and biology, and is foundational to fields such as biochemistry and material science. Chemistry is the study of matter and the transformations it undergoes.
Matter: Anything that has mass and occupies space.
Transformation: Any change in matter, including both physical and chemical changes.
Classification of Matter
Types of Matter
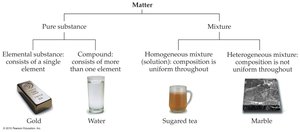
Chemistry classifies matter into two broad categories: pure substances and mixtures.
Pure Substance: Contains only a single type of matter (e.g., table salt, table sugar).
Mixture: Contains two or more substances intermingled (e.g., salt and sugar mixture, rocks).
Types of Mixtures
Mixtures are further divided into homogeneous and heterogeneous mixtures:
Homogeneous Mixture (Solution): Uniform composition throughout (e.g., tea with dissolved sugar).
Heterogeneous Mixture: Variable composition throughout; different samples may have different ratios of components (e.g., rocks, wood).
Examples of Matter Classification
Piece of wood: Heterogeneous mixture
Iron nail: Pure substance
Rusty iron nail: Heterogeneous mixture
Well-stirred mixture of food dye and water: Homogeneous mixture
Beeswax and candle wax mixed by hand: Heterogeneous mixture
Beeswax and candle wax melted, stirred, and solidified: Homogeneous mixture
Visual Examples: Water and Lemonade
Different types of mixtures and pure substances can be visually distinguished:
Pure water: Pure substance
Lemonade from powdered mix: Homogeneous mixture
Lemonade from freshly squeezed lemons: Heterogeneous mixture

Elements and Compounds
Elements
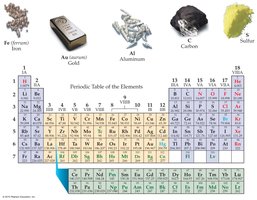
Elements are the basic building blocks of all matter and are organized in the periodic table. There are roughly 118 known elements (90 natural, 28 synthetic). Elements are represented by one-, two-, or three-letter symbols, often derived from English, Latin, or other languages.
Examples: Carbon (C), Silver (Ag), Gold (Au), Iron (Fe), Aluminum (Al)
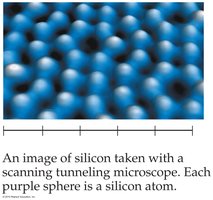
Atoms
An atom is the smallest structural unit of an element. Atoms cannot be seen with normal microscopes; the first images were obtained in the early 1980s using a scanning tunneling microscope.
Types of Pure Substances
Element: Pure substance made of only one kind of atom (e.g., pure gold is Au, oxygen in air is O2).
Compound: Pure substance made of two or more different elements (e.g., water is H2O).
Chemical Formula
A chemical formula provides the number of each type of atom that makes up the smallest unit of a compound.
Water: H2O contains two H atoms and one O atom.
Table sugar: C12H22O11 contains 12 C atoms, 22 H atoms, and 11 O atoms.
Identifying Compounds
Iron(III) oxide (Fe2O3): Compound
Ozone (O3): Not a compound (elemental form)
Iron (Fe): Not a compound (element)
Carbon monoxide (CO): Compound
Propane (C3H8): Compound
Physical Transformations of Matter
States of Matter
Matter exists in three primary states: solid, liquid, and gas. Physical transformations involve changes between these states without altering the chemical identity of the substance.
Freezing: Liquid to solid
Melting: Solid to liquid
Vaporization: Liquid to gas
Condensation: Gas to liquid
Sublimation: Solid directly to gas
Example: Ice cubes slowly vanishing in a freezer is an example of sublimation.
Chemical Transformations of Matter
Chemical Change
A chemical change (chemical transformation) occurs when a substance changes into a different substance or substances. Chemical properties are observed when a substance undergoes such changes.
Example: Combining oxygen and hydrogen gas with a spark creates water (H2O).
Chemical Reactions
A chemical reaction describes a chemical transformation. The substances at the start are called reactants, and the newly formed substances are products.
Example equation:
means "reacts to form"
States of matter: (g) = gas, (l) = liquid, (s) = solid
Identifying Chemical vs. Physical Transformations
4 P(s) + 5 O2(g) → 2 P2O5(s): Chemical transformation
H2O(g) → H2O(l): Physical transformation
3 O2(g) → 2 O3(g): Chemical transformation
