 Back
BackIntroductory Chemistry: Atomic Structure, Periodic Table, and Electron Configuration (Week 3 Study Guide)
Study Guide - Smart Notes

Models of the Atom
Historical Development of Atomic Models
The atomic model has evolved over time as scientists have gained a deeper understanding of the structure of matter. Early models were based on experimental results and theoretical reasoning.
Dalton's Model: Atoms are indivisible particles that make up elements.
Thomson's Model: Discovery of the electron led to the 'plum pudding' model.
Rutherford's Model: Atoms have a small, dense nucleus surrounded by electrons.
Bohr Model: Electrons orbit the nucleus in defined energy levels.
Example: The Bohr model explains the emission spectra of hydrogen.

Atomic Structure and Notation
Components of the Atom
Atoms consist of three main subatomic particles: protons, neutrons, and electrons. The atomic number is the number of protons in the nucleus, which determines the element.
Proton (p+): Positively charged, found in the nucleus.
Neutron (n0): Neutral, found in the nucleus.
Electron (e-): Negatively charged, orbits the nucleus.
Notation: 23Na indicates sodium with a mass number of 23.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers.
Example: Carbon-12 and Carbon-14 are isotopes of carbon.
Electron Configuration
Filling Sublevels and Orbitals
Electron configuration describes the arrangement of electrons in an atom's orbitals. Electrons fill orbitals in order of increasing energy, following the Aufbau principle, Pauli exclusion principle, and Hund's rule.
Quantum Numbers: Describe the properties of atomic orbitals and electrons.
Electron Configuration Notation: Example for sodium: 1s2 2s2 2p6 3s1
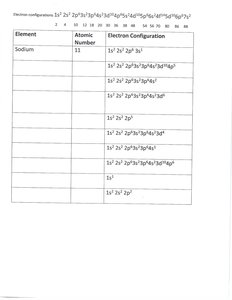
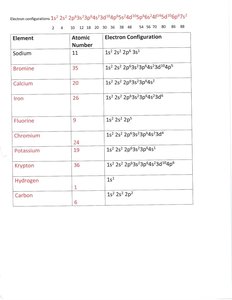
The Periodic Table
Periodic Law and Organization
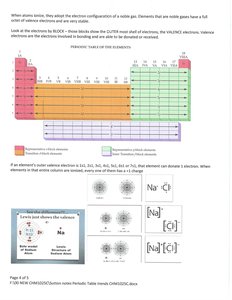
The periodic table arranges elements by increasing atomic number, revealing periodic trends in properties. Elements are grouped into families with similar chemical behaviors.
Groups: Vertical columns, elements with similar valence electron configurations.
Periods: Horizontal rows, elements with the same number of electron shells.
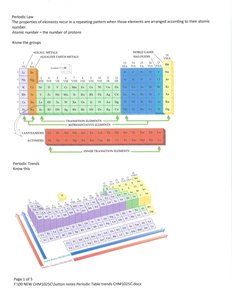
Periodic Trends
Periodic trends include atomic radius, ionization energy, and electron affinity. These trends help predict element behavior.
Atomic Radius: Generally decreases across a period and increases down a group.
Ionization Energy: Increases across a period and decreases down a group.
Electron Affinity: Tends to become more negative across a period.
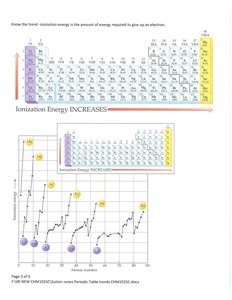
Formation of Ions and Electron Configuration
Ionization and Electron Configuration of Ions
Atoms form ions by gaining or losing electrons to achieve a stable electron configuration, often resembling that of noble gases.
Cation: Positively charged ion formed by losing electrons.
Anion: Negatively charged ion formed by gaining electrons.
Example: Sodium (Na) loses one electron to form Na+; Chlorine (Cl) gains one electron to form Cl-.

Energy Changes and Phase Transitions
Heating Curve and Phase Changes
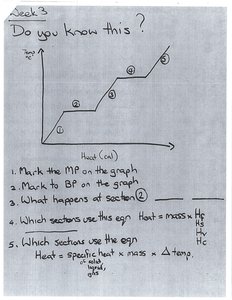
A heating curve shows the temperature change of a substance as heat is added, illustrating phase transitions such as melting and boiling.
Melting Point (MP): Temperature at which a solid becomes a liquid.
Boiling Point (BP): Temperature at which a liquid becomes a gas.
Heat Calculations:
Phase Change Calculations: (fusion), (vaporization)
Stoichiometry and Chemical Equations
Stoichiometric Calculations
Stoichiometry involves using balanced chemical equations to calculate the amounts of reactants and products in a chemical reaction.
Balanced Equation Example:
Conversion Factors: Used to relate grams, moles, and molecules.
Example Calculation: Given mass of propane, calculate moles and then volume of CO2 produced.

Study Guide and Exam Preparation
Key Concepts for Test 1
Review the following topics for comprehensive understanding and exam success:
Steps in the scientific method
Significant figures and calculation rules
Conversion factor problems (simple and complex)
Density calculations
Atomic structure and isotopes
Calculation of atomic mass unit (amu)
Electron configuration and quantum numbers
Names and symbols of elements
Percent mass calculations

Additional Info
Some images and notes reference conversion factor problems and barter systems (e.g., zebras for koalas), which are useful for practicing dimensional analysis and logical problem solving, but are not directly related to core chemistry content. Focus on chemical equations, atomic structure, and periodic trends for exam preparation.
