 Back
BackIntroductory Chemistry: Comprehensive Final Exam Review Notes
Study Guide - Smart Notes

The Chemical World
The Value of Chemistry and Problem Solving
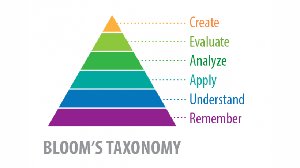
Chemistry provides a framework for understanding the natural world and developing critical problem-solving skills. In the modern workforce, foundational knowledge is essential, but higher-level skills such as analysis, evaluation, and creation are increasingly valuable.
The Scientific Method
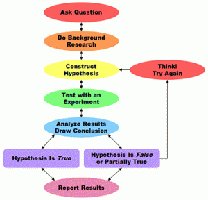
The scientific method is a systematic approach to learning about the world through observation and experimentation. It involves making observations, forming hypotheses, conducting experiments, and analyzing results to draw conclusions.
Observation: Gathering information using the senses or instruments.
Hypothesis: A tentative explanation for an observation.
Experiment: A controlled procedure to test a hypothesis.
Conclusion: A decision based on the analysis of experimental data.
Matter and Energy
Classification of Matter by Composition
Matter can be classified based on its composition into elements, molecules, compounds, and mixtures.
Element: A substance composed of only one type of atom.
Molecule: Two or more atoms chemically bonded together (e.g., O2, CO2).
Compound: A substance composed of two or more elements in fixed proportions.
Homogeneous mixture: Uniform composition throughout (e.g., saltwater).
Heterogeneous mixture: Non-uniform composition (e.g., salad).

Physical and Chemical Properties and Changes
Properties and changes of matter are classified as physical or chemical.
Physical property: Observed without changing composition (e.g., color, boiling point).
Chemical property: Observed only by changing composition (e.g., flammability).
Physical change: Alters form, not composition (e.g., melting, dissolving).
Chemical change: Produces new substances (e.g., burning, rusting).
Measurement and Problem Solving
Significant Figures in Measurement
Significant figures reflect the precision of a measurement. The last digit is always an estimate, and measurements should always be recorded one place beyond the smallest marked unit.

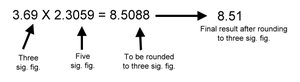
Significant Figures in Calculations
Multiplication/Division: The result has the same number of significant figures as the factor with the fewest significant figures.
Addition/Subtraction: The result has the same number of decimal places as the quantity with the fewest decimal places.


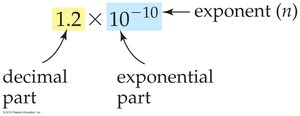
Scientific Notation
Scientific notation expresses very large or small numbers as a product of a decimal part and an exponential part (power of ten). All digits in the decimal part are significant.
Example: $1.2 \times 10^{-10}$

SI (Metric) Units and Prefixes
The metric system uses base units and prefixes to indicate multiples or fractions of units. Common prefixes include kilo-, centi-, and milli-.
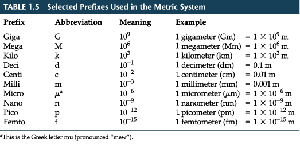

Unit (Dimensional) Analysis
Unit analysis is a method for solving problems by converting between units using conversion factors. Always start with the given value and apply conversion factors until the desired unit is reached.

Chemical Composition
Density
Density is the ratio of mass to volume and is used to identify substances and solve conversion problems.
Formula: $\text{Density} = \frac{\text{Mass}}{\text{Volume}}$

Atoms and Elements
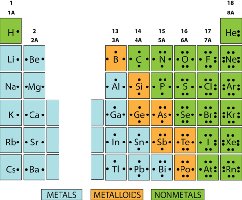
Atomic Notation
Atomic number is the number of protons, mass number is protons plus neutrons, and charge is protons minus electrons. The atomic number determines the element's identity.
Valence Electrons and Ions
Valence electrons are the outermost electrons and determine chemical properties. Atoms gain or lose electrons to form ions and achieve a stable electron configuration (octet rule).
Cation: Positive ion (lost electrons)
Anion: Negative ion (gained electrons)
Electron Configuration
Electrons fill atomic orbitals in a specific order, which can be determined using the periodic table. Ions have the same electron configuration as the nearest noble gas.
Chemical Reactions
Balancing Chemical Equations
Balancing equations ensures the same number of each atom on both sides of the reaction. Use coefficients to balance atoms, starting with the most complex molecule.
Types of Chemical Reactions
Single Displacement: One element replaces another in a compound.
Double Displacement: Exchange of ions between two compounds.
Combustion: Reaction with oxygen producing energy, CO2, and H2O.


Quantities in Chemical Reactions
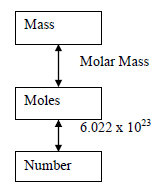
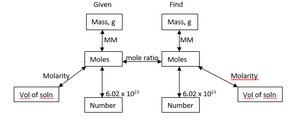
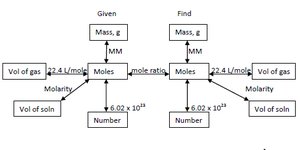
Mole Concept and Conversions
The mole is a counting unit for atoms and molecules. Use Avogadro's number ($6.022 \times 10^{23}$) for conversions between moles and number of particles.
Percent Composition and Empirical Formulas
Percent composition is the mass percent of each element in a compound. The empirical formula is the simplest whole-number ratio of elements in a compound.

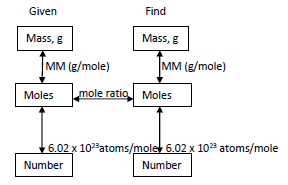
Stoichiometry
Stoichiometry involves using balanced equations to calculate quantities of reactants and products. Steps include converting to moles, applying mole ratios, and converting to desired units.
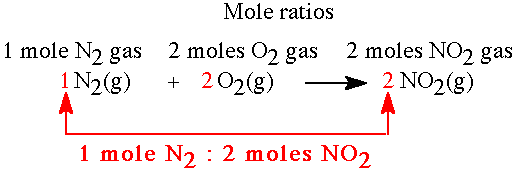
Theoretical Yield, Actual Yield, and Percent Yield
Theoretical yield is the calculated maximum amount of product, actual yield is what is obtained, and percent yield is the ratio of actual to theoretical yield times 100%.

Chemical Bonding
Lewis Dot Structures
Lewis dot diagrams represent valence electrons and help visualize bonding in molecules. Follow a stepwise process to draw structures and identify resonance if multiple valid structures exist.
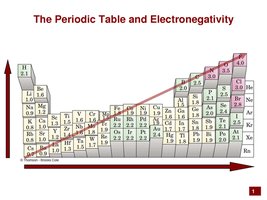
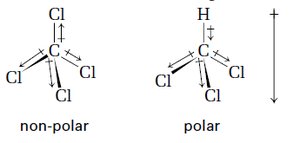
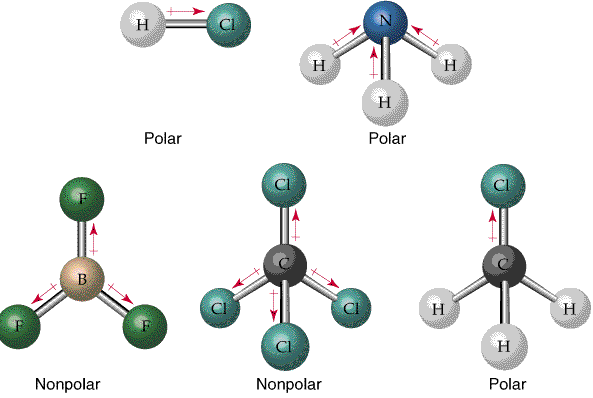
Polarity of Bonds and Molecules
Bonds are polar if there is a significant difference in electronegativity between atoms. Molecular polarity depends on both bond polarity and molecular shape.
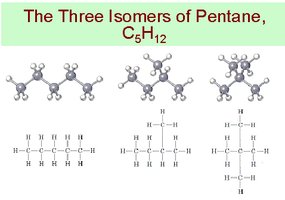
Isomers
Isomers are compounds with the same molecular formula but different structures and properties.
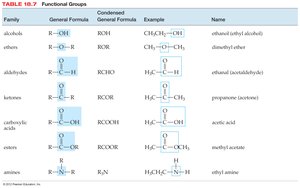
Naming Hydrocarbons and Functional Groups
Hydrocarbons are named based on the longest carbon chain and the presence of branches or multiple bonds. Functional groups determine the chemical properties of organic molecules.
Liquids, Solids, and Solutions
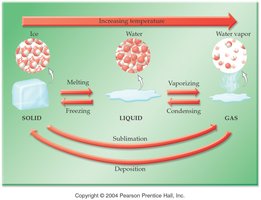
Phase Changes
Phase changes include melting, freezing, vaporization, condensation, sublimation, and deposition. These changes involve energy transfer but not chemical composition.
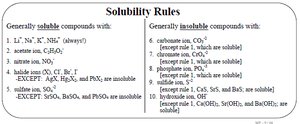
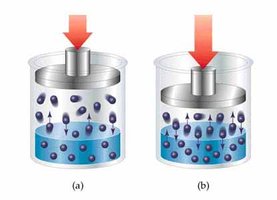
Types of Solutions and Solubility
Solubility depends on the nature of solute and solvent. Gases are more soluble at higher pressure and lower temperature. Polar and ionic compounds dissolve in water, while nonpolar compounds do not.
Measuring Concentration
Mass percent: $\frac{\text{g solute}}{\text{g solute} + \text{g solvent}} \times 100\%$
Molarity (M): $\frac{\text{moles solute}}{\text{liters solution}}$
Dilution of Solutions
When diluting solutions, the amount of solute remains constant. Use the equation $M_1V_1 = M_2V_2$ to calculate new concentrations or volumes.
Solution Stoichiometry
Use molarity and volume to find moles of solute, then apply stoichiometry to solve reaction problems.
Acids, Bases, and pH
Properties of Acids and Bases
Acids: Sour taste, dissolve metals, turn blue litmus red.
Bases: Bitter taste, slippery feel, turn red litmus blue.



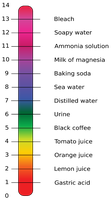
pH Scale
The pH scale measures the acidity or basicity of a solution. $\text{pH} = -\log[H^+]$. Acidic solutions have pH < 7, basic solutions have pH > 7, and neutral solutions have pH = 7.
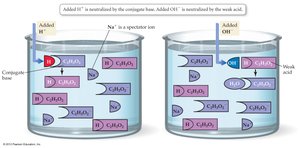
Buffers
Buffers are solutions that resist changes in pH. They contain a weak acid and its conjugate base, which neutralize added acids or bases.
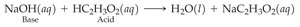
Gases
Kinetic Molecular Theory and Properties of Gases
Gases have indefinite shape and volume, low density, and are compressible. The kinetic molecular theory explains these properties by describing gas particles as being in constant, random motion with negligible attractions between them.
Ideal Gas Law
The ideal gas law relates pressure, volume, temperature, and amount of gas: $PV = nRT$. R is the gas constant (0.0821 L·atm/mol·K). Temperature must be in Kelvin.

Gas Stoichiometry
At standard temperature and pressure (STP), 1 mole of any gas occupies 22.4 L. Use this relationship for stoichiometry involving gases.
Radioactivity and Nuclear Chemistry
Types of Radioactive Decay
Radioactive decay includes alpha emission, beta emission, and positron emission. Each type changes the nucleus and produces a different daughter nuclide.
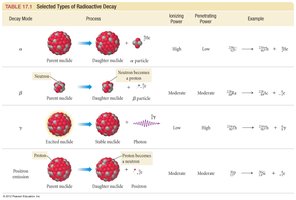
Half-Life Calculations
The half-life of a radioactive isotope is the time required for half of a sample to decay. Use a table to track the amount remaining after each half-life interval.
Additional info: These notes provide a comprehensive review of key concepts in introductory chemistry, including definitions, examples, and problem-solving strategies. For more detailed practice, refer to your textbook and assigned homework problems.
