 Back
BackIntroductory Chemistry Exam 1 – Step-by-Step Study Guidance
Study Guide - Smart Notes

Q4. What is the density of the solid object that is weighed and submerged in water?
Background
Topic: Density and Measurement
This question tests your understanding of how to determine the density of an object using its mass and the volume of water it displaces (water displacement method).
Key Terms and Formulas
Density (): The mass of a substance per unit volume.
Formula:
Water Displacement: The volume of water displaced by the object equals the volume of the object.
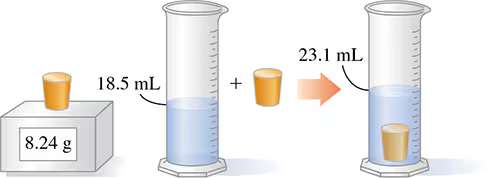
Step-by-Step Guidance
Identify the mass of the object from the balance. In the image, the mass is given as 8.24 g.
Determine the initial volume of water in the graduated cylinder (before the object is added). The image shows 18.5 mL.
Determine the final volume of water after the object is submerged. The image shows 23.1 mL.
Calculate the volume of the object by subtracting the initial volume from the final volume: .
Set up the density formula using the mass and the calculated volume: .
Try solving on your own before revealing the answer!
Final Answer: 1.79 g/mL
Using , then .
This value represents the density of the solid object based on its mass and the volume of water it displaced.
