 Back
BackIntroductory Chemistry Exam 1 Study Guide
Study Guide - Smart Notes

CHAPTER 1: THE CHEMICAL WORLD
Definitions and the Scientific Method
The study of chemistry involves understanding the properties, composition, and changes of matter. The scientific method is a systematic approach used to investigate natural phenomena, develop hypotheses, and build scientific knowledge.
Chemistry: The science that studies the composition, structure, properties, and changes of matter.
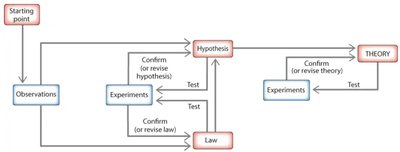
Scientific Method: A logical, systematic process for investigating phenomena, acquiring new knowledge, or correcting previous knowledge.
Key Steps: Observations, hypothesis formation, experimentation, and development of laws and theories.
Hypothesis: A tentative explanation for an observation, which can be tested by experiments.
Experiment: A controlled procedure to test the validity of a hypothesis.
Scientific Law: A statement that summarizes past observations and predicts future ones; describes what happens but not why.
Scientific Theory: A well-substantiated explanation of some aspect of the natural world; explains why phenomena occur.
Law vs. Theory: Laws describe what happens; theories explain why it happens.
CHAPTER 2: MEASUREMENT AND PROBLEM SOLVING
Scientific Notation
Scientific notation is used to express very large or very small numbers in a compact form. It consists of a decimal part and an exponential part.
Decimal Part: Usually between 1 and 10.
Exponential Part: 10 raised to an exponent n ().
Positive Exponent: Indicates multiplication by 10 n times (e.g., ).
Negative Exponent: Indicates division by 10 n times (e.g., ).
Example: 0.000038 in scientific notation is .
Example: is 5,219.
Significant Figures
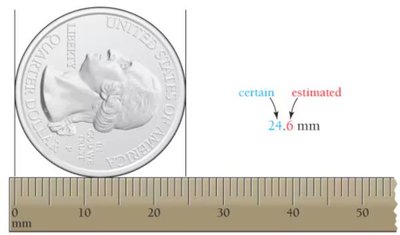
Significant figures reflect the precision of a measurement. The number of significant digits indicates the certainty of the measurement.
Precision: More digits = more precision; fewer digits = less precision.
Estimated Digit: The last digit is estimated and indicates uncertainty.
Reporting Measurements: Only report digits that can be measured with certainty plus one estimated digit.
Example: Measuring a quarter with a ruler marked every 1 mm yields 24.6 mm (the 6 is estimated).
Rules for Counting Significant Figures
All nonzero digits are significant (e.g., 1.243 has 4 significant figures).
Interior zeros (between nonzero digits) are significant (e.g., 1.204 has 4 significant figures).
Trailing zeros after a decimal point are significant (e.g., 5.100 has 4 significant figures).
Trailing zeros before a decimal point are significant (e.g., 100.3 has 4 significant figures).
Leading zeros are not significant (e.g., 0.005 has 1 significant figure).
Trailing zeros at the end of a number without a decimal point are ambiguous (e.g., 3,200 could have 2 or 4 significant figures).
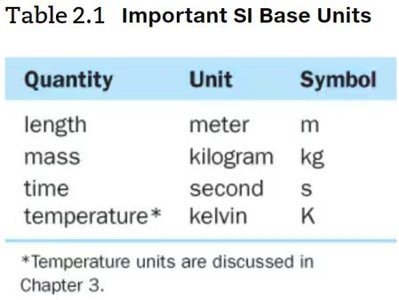
SI Units and Prefix Multipliers
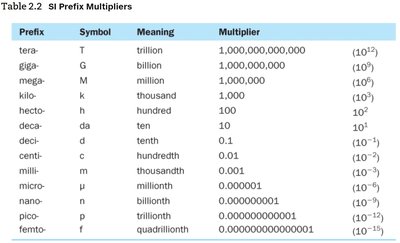
The International System of Units (SI) is a standardized system used globally. Prefix multipliers change the value of base units by powers of ten.
Base Units: Meter (m), kilogram (kg), second (s), kelvin (K).
Prefix Multipliers: Used to express multiples or fractions of base units (e.g., milli = , kilo = ).
Prefix | Symbol | Meaning | Multiplier |
|---|---|---|---|
kilo | k | thousand | 1,000 () |
milli | m | thousandth | 0.001 () |
micro | μ | millionth | 0.000001 () |
centi | c | hundredth | 0.01 () |
mega | M | million | 1,000,000 () |
nano | n | billionth | 0.000000001 () |
Additional info: | See full table for more prefixes. |
Density
Density is the ratio of mass to volume and is a fundamental property of matter.
Formula:
If mass remains constant and volume increases, density decreases.
If mass remains constant and volume decreases, density increases.
CHAPTER 3: MATTER AND ENERGY
Definitions: Matter, Atoms, Molecules, States of Matter
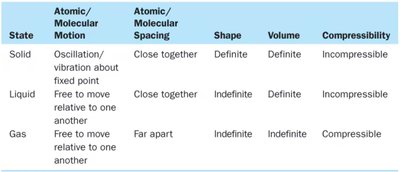
Matter is anything that has mass and occupies space. Atoms are the basic units of matter, and molecules are groups of atoms bonded together. Matter exists in three primary states: solid, liquid, and gas.
Solid: Atoms/molecules are packed closely in fixed locations; definite shape and volume; can be crystalline or amorphous.
Liquid: Atoms/molecules are close but free to move; indefinite shape, definite volume.
Gas: Atoms/molecules are far apart and free to move; indefinite shape and volume; compressible.
State | Atomic/Molecular Motion | Atomic/Molecular Spacing | Shape | Volume | Compressibility |
|---|---|---|---|---|---|
Solid | Oscillation/vibration about fixed point | Close together | Definite | Definite | Incompressible |
Liquid | Free to move relative to one another | Close together | Indefinite | Definite | Incompressible |
Gas | Free to move relative to one another | Far apart | Indefinite | Indefinite | Compressible |
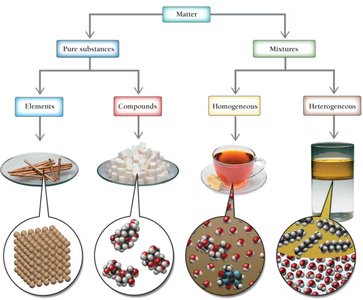
Classification of Matter
Matter can be classified as pure substances or mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Pure Substance: Composed of only one type of atom or molecule.
Element: A substance that cannot be broken down into simpler substances.
Compound: A substance composed of two or more elements in fixed proportions.
Mixture: Composed of two or more substances.
Homogeneous Mixture: Uniform composition throughout (e.g., tea).
Heterogeneous Mixture: Non-uniform composition (e.g., oil and water).
Solution: A homogeneous mixture.
Properties and Changes in Matter
Physical Property: Observed without changing composition (e.g., odor, color, melting point).
Chemical Property: Observed only when composition changes (e.g., flammability, acidity).
Physical Change: Alters appearance, not composition (e.g., melting ice).
Chemical Change: Alters composition (e.g., rusting iron).
Chemical Reaction: Process where matter undergoes a chemical change; reactants form products.
Energy and Its Forms
Energy is the capacity to do work. Work is the action of a force through a distance. Energy can be kinetic (motion) or potential (position).
Law of Conservation of Energy: Energy is neither created nor destroyed.
Kinetic Energy: Energy of motion.
Potential Energy: Energy due to position or composition.
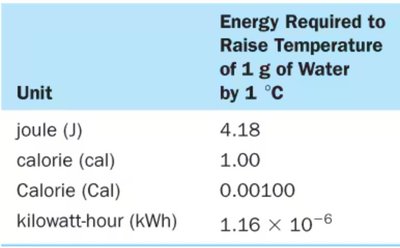
Units of Energy: Joule (J), calorie (cal), kilowatt-hour (kWh).
Exothermic Reaction: Releases energy.
Endothermic Reaction: Absorbs energy.
Temperature Scales
Temperature measures thermal energy. Common scales are Fahrenheit (°F), Celsius (°C), and Kelvin (K).
Absolute Zero: Lowest possible temperature (0 K).
Freezing Point of Water: 0°C, 32°F, 273.15 K.
Boiling Point of Water: 100°C, 212°F, 373.15 K.
CHAPTER 4: ATOMS AND ELEMENTS
Dalton’s Atomic Theory
Dalton’s atomic theory explains the nature of matter and the behavior of atoms.
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Structure of the Atom
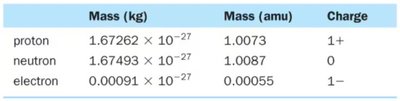
Atoms consist of protons, neutrons, and electrons. The nucleus contains protons and neutrons; electrons are dispersed in the surrounding space.
Proton: Positively charged particle in the nucleus.
Neutron: Neutral particle in the nucleus.
Electron: Negatively charged particle outside the nucleus.
Particle | Mass (kg) | Mass (amu) | Charge |
|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.0073 | 1+ |
Neutron | 1.67493 × 10-27 | 1.0087 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | 1– |
Atomic Number and the Periodic Table
The atomic number (Z) is the number of protons in an atom’s nucleus and defines the element. The periodic table arranges elements by atomic number.
Chemical Symbol: One- or two-letter abbreviation for each element.
Periodic Law: Properties of elements recur periodically when arranged by increasing atomic mass.
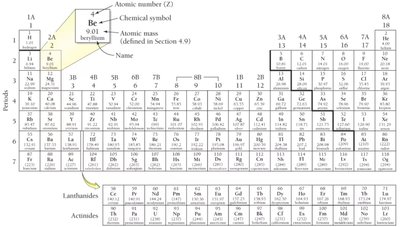
Classification of Elements
Metals: Left side; good conductors, malleable, ductile, tend to lose electrons.
Nonmetals: Upper right; varied properties, poor conductors, tend to gain electrons.
Metalloids: Along zigzag line; mixed properties, semiconductors.
Groups/Families: Columns with similar properties (e.g., alkali metals, halogens, noble gases).
Ions and Isotopes
Ions: Charged particles formed when atoms gain or lose electrons.
Anion: Negatively charged ion (gains electrons).
Cation: Positively charged ion (loses electrons).
Isotope: Atoms of the same element with different numbers of neutrons.
Additional info: Calculating atomic mass is not required for Exam 1.
