 Back
BackIntroductory Chemistry Final Exam Study Guidance
Study Guide - Smart Notes

Q15. Which orbital-filling diagram represents the ground state of oxygen?
Background
Topic: Electron Configuration and Orbital-Filling Diagrams
This question tests your understanding of how electrons fill atomic orbitals according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle. You need to identify which diagram correctly represents the ground state (lowest energy) electron configuration for an oxygen atom.
Key Terms and Concepts:
Ground State: The lowest energy arrangement of electrons in an atom.
Aufbau Principle: Electrons fill orbitals starting with the lowest energy first.
Hund's Rule: Every orbital in a subshell is singly occupied before any orbital is doubly occupied.
Pauli Exclusion Principle: No two electrons in the same atom can have the same set of four quantum numbers (each orbital can hold a maximum of two electrons with opposite spins).
Oxygen Atomic Number: 8 (so 8 electrons to place in orbitals).
Step-by-Step Guidance
Recall the order in which orbitals are filled for the first 8 electrons: .
Write the electron configuration for oxygen: .
Translate this configuration into an orbital-filling diagram. For the subshell, remember Hund's rule: fill each orbital with one electron before pairing.
Check each diagram for the correct number of electrons and proper application of Hund's rule and the Pauli exclusion principle.
Try solving on your own before revealing the answer!
Final Answer:
The correct orbital-filling diagram for the ground state of oxygen is the fourth image (bottom right):
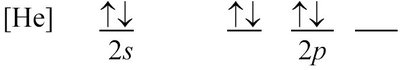
This diagram shows two electrons in the orbital and four electrons in the orbitals, with each orbital singly occupied before any pairing, consistent with Hund's rule.
