 Back
BackIntroductory Chemistry: Matter, Properties, and Separation Techniques
Study Guide - Smart Notes

Matter and Its Classification
Definition and Types of Matter
Matter is anything that occupies space and has mass. It can be classified based on its composition and physical state. The two main categories are mixtures and pure substances.
Mixtures: Physical combinations of two or more substances where each retains its own properties.
Pure Substances: Matter with a uniform and unchanging composition, including elements and compounds.
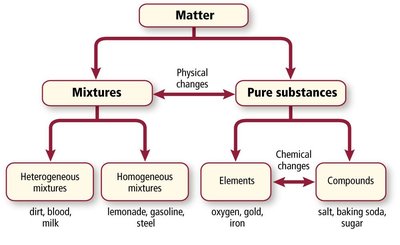
Mixtures
Heterogeneous Mixtures: Composition is not uniform throughout (e.g., dirt, blood, milk).
Homogeneous Mixtures (Solutions): Composition is uniform throughout (e.g., lemonade, gasoline, steel).
Pure Substances
Elements: Substances that cannot be broken down by chemical means (e.g., oxygen, gold, iron).
Compounds: Substances composed of two or more elements chemically combined in a fixed ratio (e.g., salt, baking soda, sugar).
Physical and Chemical Properties of Matter
Physical Properties
Physical properties can be observed or measured without changing the composition of the substance. Examples include color, density, melting point, and boiling point.
Color
Density
Melting point
Boiling point
Malleability
Ductility
Conductivity
Chemical Properties
Chemical properties describe a substance's ability to undergo changes that transform it into different substances. Examples include reactivity with acids, formation of new compounds, and changes in color due to chemical reactions.

Separation of Mixtures
Filtration
Filtration separates mixtures based on particle size. Smaller particles pass through a filter, while larger particles are retained. This technique is commonly used to separate solids from liquids.

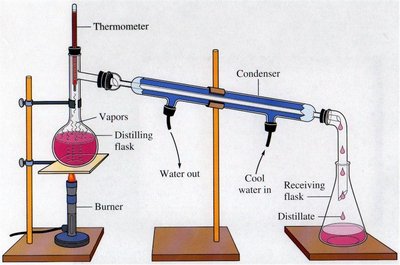
Distillation
Distillation separates substances based on differences in boiling points. The mixture is heated until one component vaporizes, then the vapor is condensed and collected separately.
Crystallization
Crystallization separates a pure solid from a solution. As the solvent evaporates, the solute forms crystals. This method is used for substances like salt or sugar from water solutions.

Chromatography
Chromatography separates mixtures based on the affinity of substances for a stationary phase versus a mobile phase. Components move at different rates, resulting in separation.

Particle Diagrams and Models
Visualizing Elements, Compounds, and Mixtures
Particle diagrams are used to represent the arrangement of particles in elements, compounds, and mixtures. In these diagrams, each sphere represents an atom or molecule. The arrangement and uniformity help distinguish between pure substances and mixtures.
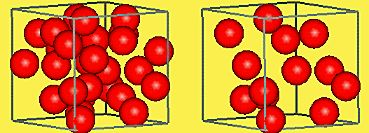
Left: Homogeneous mixture (even distribution). Right: Heterogeneous mixture (uneven distribution).
Summary Table: Properties of Copper
Physical Properties | Chemical Properties |
|---|---|
|
|
