 Back
BackIntroductory Chemistry: Measurement, Matter, and Atomic Structure Study Guide
Study Guide - Smart Notes

Chapter 2: Measurement and Problem Solving
Key Vocabulary and Concepts
Exact Number: A value known with complete certainty, often from counting or defined quantities (e.g., 1 dozen = 12).
Measurement: A quantitative observation that includes both a number and a unit (e.g., 25.0 mL).
Precision: The degree to which repeated measurements under unchanged conditions show the same results.
Accuracy: How close a measured value is to the true or accepted value.
Significant Figures (Sig Figs): The digits in a measurement that are known with certainty plus one estimated digit.
SI Units: The International System of Units, the standard units used in science (e.g., meter, kilogram, second).
Mass: The amount of matter in an object, measured in kilograms (kg).
Volume: The amount of space an object occupies, measured in liters (L) or cubic centimeters (cm3).
Density: The mass per unit volume of a substance, calculated as .
Unit Analysis (Dimensional Analysis): A method for converting between units using conversion factors.
Conversion Factor: A ratio used to express a quantity in different units.
Uncertainty: The degree of doubt in a measurement, often indicated by the last significant figure.
Applying Accuracy and Precision
Accuracy refers to how close a measurement is to the true value; precision refers to how close repeated measurements are to each other.
Example: If you weigh a 10.00 g standard mass and obtain values of 9.98 g, 9.99 g, and 10.01 g, your measurements are both accurate and precise.
Scientific Notation
Used to express very large or very small numbers in the form .
Example: 0.00045 =
Significant Figures in Calculations
Addition/Subtraction: The result should have the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: The result should have the same number of significant figures as the measurement with the fewest significant figures.
Ambiguous Zeros: Zeros between nonzero digits are significant; leading zeros are not; trailing zeros are significant only if there is a decimal point.
Rounding Calculated Values
Round the final answer to the correct number of significant figures based on the rules above.
Unit Conversions
Use conversion factors to change from one unit to another (e.g., inches to centimeters).
Metric prefixes (kilo-, centi-, milli-, etc.) are used to indicate multiples or fractions of units.
Area and volume conversions often require squaring or cubing the conversion factor.

Density Calculations
Density is calculated as .
Density can be used as a conversion factor between mass and volume.
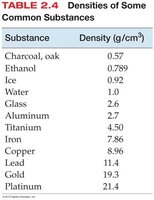
SI Units for Common Quantities
Mass: kilogram (kg)
Volume: liter (L)
Time: second (s)
Temperature: kelvin (K)
Chapter 3: Matter and Energy
Classification of Matter
Matter: Anything that has mass and occupies space.
Pure Substance: Matter with a fixed composition (element or compound).
Mixture: A combination of two or more substances that are not chemically combined.
Homogeneous Mixture (Solution): Uniform composition throughout (e.g., saltwater).
Heterogeneous Mixture: Non-uniform composition (e.g., salad, sand and iron filings).
Element: A pure substance made of only one kind of atom.
Compound: A pure substance composed of two or more elements chemically combined.
Physical and Chemical Properties and Changes
Physical Properties: Characteristics that can be observed without changing the substance (e.g., color, melting point).
Physical Change: A change that does not alter the chemical composition (e.g., melting, boiling).
Chemical Properties: Characteristics that describe a substance's ability to undergo a chemical change (e.g., flammability).
Chemical Change: A change that produces one or more new substances (e.g., rusting, burning).
Visible signs of chemical change: color change, gas production, precipitate formation, temperature change.
Law of Conservation of Mass
Mass is neither created nor destroyed in a chemical reaction; the total mass of reactants equals the total mass of products.
Energy and Its Forms
Energy: The capacity to do work or produce heat.
Kinetic Energy: Energy of motion.
Potential Energy: Stored energy due to position or composition.
Other forms: electrical, thermal, chemical energy.
Energy Units and Conversions
Common units: joule (J), calorie (cal), kilowatt-hour (kWh).
1 calorie (cal) = 4.184 joules (J)
1 Calorie (Cal) = 1000 calories (cal)
1 kilowatt-hour (kWh) = joules (J)

Exothermic vs. Endothermic Processes
Exothermic: Processes that release energy (heat flows out of the system).
Endothermic: Processes that absorb energy (heat flows into the system).
Temperature and Particle Motion
Temperature is a measure of the average kinetic energy of particles in a substance.
As temperature increases, particle motion increases.
Temperature Scales and Conversions
Celsius (°C), Fahrenheit (°F), Kelvin (K)
Conversions:
Specific Heat Capacity
Specific heat capacity (C) is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C.
Equation:
Where = heat (J), = mass (g), = specific heat capacity (J/g·°C), = change in temperature (°C).
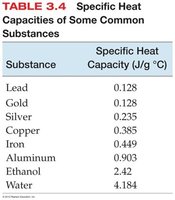
Chapter 4: Atoms and Elements
Key Vocabulary
Electron: Negatively charged subatomic particle found outside the nucleus.
Proton: Positively charged subatomic particle found in the nucleus.
Neutron: Neutral subatomic particle found in the nucleus.
Nucleus: Dense center of the atom containing protons and neutrons.
Atomic Number (Z): Number of protons in the nucleus; defines the element.
Mass Number (A): Total number of protons and neutrons in the nucleus.
Atomic Mass: Weighted average mass of the atoms in a naturally occurring element.
Atomic Mass Unit (amu): Standard unit for atomic mass; 1 amu = 1/12 the mass of a carbon-12 atom.
Isotopes: Atoms of the same element with different numbers of neutrons.
Ions: Atoms or molecules with a net electric charge due to loss or gain of electrons.
Cation: Positively charged ion (loss of electrons).
Anion: Negatively charged ion (gain of electrons).
Periodicity: The recurring trends in properties of elements across periods of the periodic table.
Group: Vertical column in the periodic table.
Period: Horizontal row in the periodic table.
Transition Metals: Elements in groups 3-12.
Lanthanide and Actinide Series: Two rows below the main body of the periodic table.
Noble Gases: Group 18 elements, inert gases.
Metals, Nonmetals, Metalloids: Classification based on properties and location in the periodic table.
Alkali Metals: Group 1 elements.
Alkaline Earth Metals: Group 2 elements.
Chalcogens: Group 16 elements.
Halogens: Group 17 elements.
Law of Conservation of Mass in Chemical Reactions
The total mass of reactants equals the total mass of products in a chemical reaction.
Isotopes and Atomic Structure
Isotopes are identified by their mass number (e.g., carbon-12, carbon-14).
Number of protons = atomic number; number of neutrons = mass number - atomic number; number of electrons = number of protons (for neutral atoms).
Ions and Their Charges
Main group elements tend to form ions with predictable charges (e.g., Group 1 forms +1, Group 17 forms -1).
Cations are formed by losing electrons; anions are formed by gaining electrons.
Classification of Elements
Representative (main group) elements: Groups 1, 2, and 13-18.
Transition elements: Groups 3-12.
Metals: Left and center of the periodic table; good conductors, malleable, ductile.
Nonmetals: Right side; poor conductors, brittle.
Metalloids: Properties intermediate between metals and nonmetals; found along the stair-step line.
Calculating Atomic Mass from Isotopic Abundances
Atomic mass is calculated as the weighted average of the masses of all naturally occurring isotopes.
Equation:
Essential Memorization
Element names and symbols for elements #1-56 and 72-86.
Metric prefixes, symbols, and exponents.
Tables for Reference
Common unit conversions for length, mass, and volume.
Densities of common substances.
Energy conversion factors.
Specific heat capacities of common substances.
Additional info: Students should be able to interpret and analyze data from tables and graphs, as well as apply these concepts to solve quantitative problems in chemistry.
