 Back
BackIntroductory Chemistry: Measurement, Scientific Notation, and Problem Solving
Study Guide - Smart Notes

Chemistry in Our Lives
Chemistry and Chemicals
Chemistry is the science that studies matter, its composition, structure, properties, and the reactions it undergoes. Matter is anything that has mass and occupies space. Chemistry is divided into two main branches: inorganic chemistry (substances not containing carbon) and organic chemistry (substances containing carbon).
Chemical (or substance): A material with a definite composition and properties.
Example: Toothpaste contains chemicals such as calcium carbonate (abrasive), sorbitol (prevents hardening), sodium lauryl sulfate (loosens plaque), titanium dioxide (whitens), sodium fluorophosphate (strengthens enamel), and methyl salicylate (flavor).

Chemistry and Measurements
Units of Measurement
The metric system (SI) is a decimal system of units used internationally in science. It is based on standard units and uses prefixes to express quantities larger or smaller than the base unit.
Volume: SI unit is cubic meter (m3), common units are liter (L) and milliliter (mL).
Length: SI unit is meter (m).
Mass: SI unit is kilogram (kg).
Temperature: SI unit is kelvin (K), also degrees Celsius (°C) and Fahrenheit (°F).
Time: SI unit is second (s).
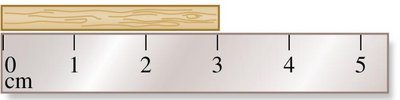
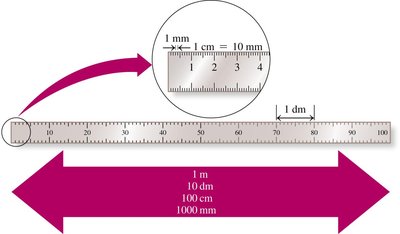
Length
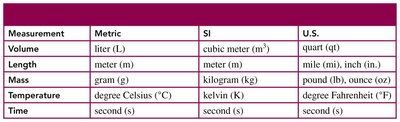
Length is measured in meters. 1 meter is slightly longer than a yard (1 m = 39.37 inches). Tools for measuring length include rulers, meter sticks, and tape measures.
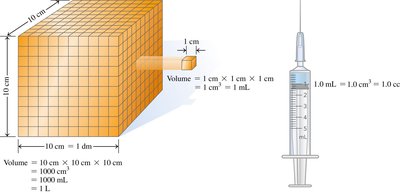
Volume
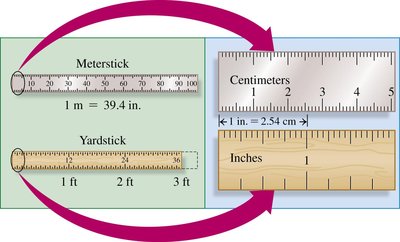
Volume is the amount of space occupied by matter. Common laboratory tools for measuring volume include graduated cylinders, beakers, pipettes, measuring cups, burets, and syringes. 1 L = 1000 mL; 1 mL = 1 cm3 = 1 cc.
Mass
Mass is the measure of the quantity of material in an object. The SI unit is kilogram (kg). 1 kg = 2.205 pounds; 1 kg = 1000 g; 454 g = 1 lb. Mass is measured using scales and balances.

Temperature
Temperature measures how hot or cold something is. The SI unit is kelvin (K), but degrees Celsius (°C) and Fahrenheit (°F) are also used. The conversion between Celsius and Kelvin is: .

Time
Time is measured in seconds (s), the SI unit. Other units include minutes, hours, days, and years. Tools for measuring time include stopwatches, clocks, sundials, and cell phones.

Measurement and Problem Solving
Scientific Notation
Scientific notation is used to represent very large or very small numbers efficiently. It consists of a coefficient, a power of 10, and a unit. The decimal point is placed to the right of the first nonzero digit.
Example: 0.00000000000095 g = g
Example: 60,000,000,000,000 cells = cells

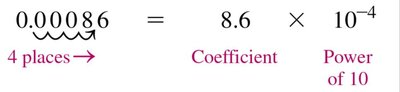

Measured Numbers and Significant Figures
Measured numbers are obtained from measuring a quantity. Significant figures include all digits observed plus one estimated digit. Rules for significant figures:
All nonzero digits are significant.
Zeros between nonzero digits (sandwiched) are significant.
Zeros after nonzero digits with a decimal point are significant.
Zeros before nonzero digits (placeholders) are not significant.
Zeros after nonzero digits without a decimal point are not significant.


Exact Numbers
Exact numbers are not measured but counted or defined by equalities. They have an unlimited number of significant figures and are not used to determine significant figures in calculations.
Examples: 12 bagels (counted), 1 L = 1000 mL (defined equality), 1 kg = 1000 g (defined equality).
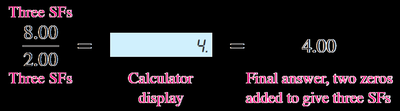
Significant Figures in Calculations
When performing calculations, the precision of the answer is limited by the least precise measurement.
Multiplication and Division: The answer must have the same number of significant figures as the measurement with the least number of significant figures.
Addition and Subtraction: The answer must be rounded to the same number of decimal places as the value with the fewest decimal places.
Rounding: If the first digit to be dropped is ≤ 4, drop it. If it is ≥ 5, increase the last retained digit by 1.
Prefixes and Equalities
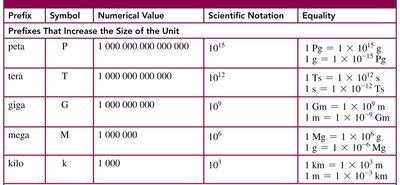
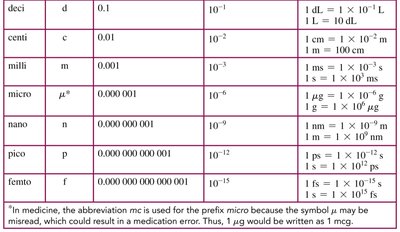
Common Prefixes for SI Units
Prefixes are used to indicate multiples or fractions of base units in the metric system.
Prefix | Symbol | Value | Scientific Notation |
|---|---|---|---|
kilo | k | 1,000 | |
centi | c | 0.01 | |
milli | m | 0.001 | |
micro | μ | 0.000001 | |
nano | n | 0.000000001 |
Equalities and Conversion Factors
Equalities express relationships between units and are used to create conversion factors for dimensional analysis.
Examples: 1 kg = 1000 g, 1 m = 1000 mm, 1 L = 1000 mL, 454 g = 1 lb.
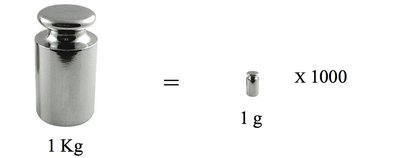

Problem Solving and Dimensional Analysis
Basic Steps to Problem Solving
Problem solving in chemistry often involves unit conversions using dimensional analysis. The steps are:
Read the problem carefully and identify known and unknown quantities.
Determine the principles and unit relationships needed.
Set up the problem using conversion factors so unwanted units cancel.
Perform the necessary mathematical operations.
Check the answer for reasonableness.
Dimensional Analysis
Dimensional analysis is a method for converting one unit to another using conversion factors. The conversion factor must cancel the given unit and introduce the needed unit.
Formula: Given unit × conversion factor(s) = Needed unit
Density and Specific Gravity
Density
Density is the ratio of the mass of a substance to the volume it occupies. It is a useful property for identifying substances and solving conversion problems.
Formula:
Units: g/mL or g/cm3
Specific Gravity
Specific gravity relates the density of a substance to the density of water (1 g/mL). It is a unitless quantity.
Formula:
Examples and Applications
Sample Problems
Convert 175 cm to meters:
Convert 4.00 oz to grams:
Calculate density:
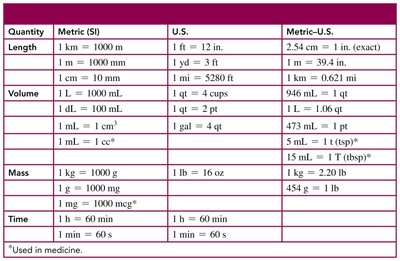
Summary Table: Common Metric Conversions
Quantity | Metric (SI) | U.S. | Metric-U.S. |
|---|---|---|---|
Length | 1 km = 1000 m | 1 ft = 12 in. | 1 in. = 2.54 cm (exact) |
Volume | 1 L = 1000 mL | 1 qt = 4 cups | 946 mL = 1 qt |
Mass | 1 kg = 1000 g | 1 lb = 16 oz | 454 g = 1 lb |
Time | 1 h = 60 min | 1 h = 60 min | 1 min = 60 s |
