 Back
BackIntroductory Chemistry: Solutions and Their Properties
Study Guide - Smart Notes

Solutions
Concept of Solutions
Solutions are homogeneous mixtures composed of two or more substances that form a uniform composition. The main components of a solution are the solute and the solvent.
Solute: The substance dissolved in the solvent, usually present in a smaller amount.
Solvent: The substance present in the largest amount, which dissolves the solute.
Concentration: A measurement of the amount of solute in a given volume of solution.
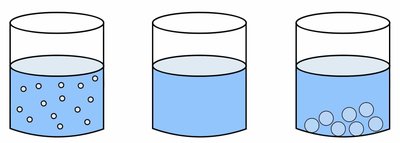
Types of Mixtures: Solutions, Suspensions, and Colloids
Mixtures can be classified based on the size of their solute particles and their behavior in the solvent.
Solutions: Particle size < 1 nm; particles do not settle; example: salt water.
Colloids: Particle size between 1 nm and 500 nm; particles do not settle; example: milk.
Suspensions: Particle size > 500 nm; particles settle on standing; example: salad dressing.
Example: If 10.0 g NaCl is dissolved in 500.0 mL of water, NaCl is the solute and water is the solvent.
Solubility and Intermolecular Forces
Solubility Concept
Solubility is determined by the nature of intermolecular forces. According to the theory of "like dissolves like," compounds with similar intermolecular forces or polarity will dissolve into each other.
Polar solutes dissolve in polar solvents.
Nonpolar solutes dissolve in nonpolar solvents.
Example: Predict whether a solution will form between CCl4 and P4 (both nonpolar), or CH3OH and C6H6 (polar and nonpolar).
Concentration Units
Mass Percent
Mass percent is the percentage of a given element or compound within a solution.
Formula:
Example: 23.0% NaOH means 23.0 grams NaOH in 100 grams of solution.
Molarity
Molarity (M) is the number of moles of solute per liter of solution.
Formula:
Example: 5.8 M NaCl means 5.8 moles NaCl in 1.0 L of solution.
Osmolarity
Concept of Osmolarity
Osmolarity is the number of moles of ions per liter of solution. It is important for solutions containing ionic compounds.
Formula:
Solubility: Temperature and Pressure Effects
Temperature Effect
The solubility of solids and gases in liquids is affected by temperature.
As temperature increases, the solubility of most solids increases.
As temperature increases, the solubility of gases decreases.
Pressure Effect (Henry's Law)
The solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid.
Henry's Law: (where is solubility, is Henry's constant, is partial pressure)

Dilutions
Concept of Dilution
To dilute a solution, more solvent is added, decreasing the concentration of solute.
Formula: (where and are initial molarity and volume, and are final molarity and volume)
Solution Stoichiometry
Stoichiometric Calculations in Solutions
Stoichiometry in solutions involves using volume and molarity to determine the amount of reactants and products.
Convert given quantity to moles.
Use mole-to-mole ratios from balanced equations.
Convert moles to desired units (grams, volume, etc.).
Molality
Concept of Molality
Molality (m) is the number of moles of solute per kilogram of solvent.
Formula:
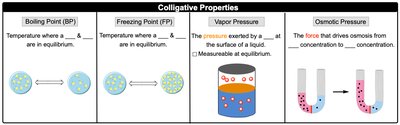
Colligative Properties
Overview of Colligative Properties
Colligative properties are physical properties of solutions that depend on the number of solute particles, not their identity. The four main colligative properties are:
Boiling Point Elevation
Freezing Point Depression
Vapor Pressure Lowering
Osmotic Pressure
Van't Hoff Factor
The Van't Hoff factor (i) is the number of particles produced from dissolving a solute.
Ionic compounds dissociate into ions; i equals the total number of ions.
Covalent compounds do not dissociate; i = 1.
Boiling Point Elevation
Concept and Formula
Adding a solute to a pure solvent increases the boiling point of the solvent.
Formula:
= change in boiling point
= Van't Hoff factor
= boiling point constant (specific to solvent)
= molality of solution
Freezing Point Depression
Concept and Formula
Adding a solute to a pure solvent decreases the freezing point of the solvent.
Formula:
= change in freezing point
= Van't Hoff factor
= freezing point constant (specific to solvent)
= molality of solution
Osmosis and Osmotic Pressure
Concept of Osmosis
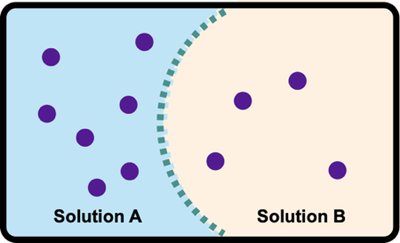
Osmosis is the net movement of solvent (usually water) across a semipermeable membrane from a region of lower solute concentration to higher solute concentration.
Semipermeable membrane: Allows solvent but not solute molecules to pass.
Osmotic pressure: The pressure exerted by the solvent moving across the membrane.
Tonicity and Effects on Cells
Concept of Tonicity
Tonicity describes the relative concentration of solutes in solutions separated by a membrane. It affects the movement of water and the health of cells.
Hypotonic: Lower solute concentration outside the cell; water enters the cell, causing swelling (hemolysis).
Isotonic: Equal solute concentration; no net movement of water.
Hypertonic: Higher solute concentration outside the cell; water leaves the cell, causing shrinkage (crenation).
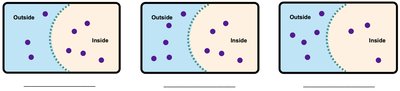
Example: A red blood cell placed in pure water will swell due to water entering the cell (hemolysis).
Additional info: These concepts are fundamental to understanding solution chemistry and its applications in biological and industrial contexts.
