 Back
BackIntroductory Chemistry Study Guide: Chapters 1–4
Study Guide - Smart Notes

Ch.1 Introduction to Chemistry
Key Concepts in Chemistry
Chemistry is the study of matter, its properties, and the changes it undergoes. Understanding chemistry involves learning about elements, compounds, mixtures, and the laws governing physical and chemical changes.
Element: A pure substance consisting of only one type of atom (e.g., Oxygen).
Compound: A substance formed from two or more elements chemically bonded (e.g., Water).
Mixture: A combination of two or more substances not chemically bonded. Homogeneous mixtures are uniform throughout (e.g., saltwater), while heterogeneous mixtures are not (e.g., sand and water).
Physical property: Can be observed without changing the substance (e.g., melting point).
Chemical property: Describes a substance's ability to undergo chemical changes (e.g., flammability).
Ch.2 The Metric System
Metric Prefixes and Unit Conversions
The metric system uses prefixes to indicate multiples or fractions of base units such as meter, gram, liter, and second. Understanding these prefixes is essential for converting between units.
Base units: meter (length), gram (mass), liter (volume), second (time).
Metric prefixes: Used to express larger or smaller quantities.
Prefix | Symbol | Multiple/Fraction |
|---|---|---|
exa- | E | 1 × 1018 |
peta- | P | 1 × 1015 |
tera- | T | 1 × 1012 |
giga- | G | 1 × 109 |
mega- | M | 1 × 106 |
kilo- | k | 1 × 103 |
deci- | d | 1 × 10-1 |
centi- | c | 1 × 10-2 |
milli- | m | 1 × 10-3 |
micro- | μ | 1 × 10-6 |
nano- | n | 1 × 10-9 |
pico- | p | 1 × 10-12 |
femto- | f | 1 × 10-15 |
atto- | a | 1 × 10-18 |
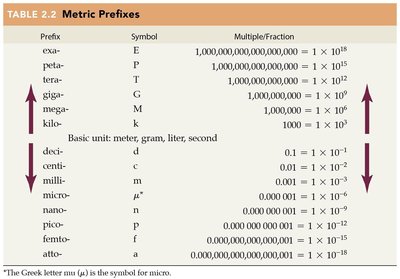
English-Metric Equivalents
Conversions between English and metric units are common in chemistry. Knowing these equivalents helps in solving problems involving measurements.
Quantity | English Unit | Metric Equivalent |
|---|---|---|
Length | 1 inch (in.) | 1 in. = 2.54 cm |
Mass | 1 pound (lb) | 1 lb = 454 g |
Volume | 1 quart (qt) | 1 qt = 946 mL |
Time | 1 second (sec) | 1 sec = 1.00 s |
Unit Conversion and Prefixes
To convert between units, use the appropriate conversion factor. For example, to convert 5 kilometers to meters:
1 kilometer = 1,000 meters
5 kilometers × 1,000 meters/kilometer = 5,000 meters
Use metric prefixes to express values in scientific notation.
Ch.3 Matter and Energy
Classification of Matter
Matter can be classified as elements, compounds, homogeneous mixtures, or heterogeneous mixtures. Understanding these classifications is fundamental to chemistry.
Element: Pure substance with only one type of atom.
Compound: Substance formed from two or more elements chemically combined.
Homogeneous mixture: Uniform composition throughout (e.g., air).
Heterogeneous mixture: Non-uniform composition (e.g., salad).
Physical and Chemical Properties
Properties of matter are classified as physical or chemical. Physical properties can be observed without changing the substance, while chemical properties involve a change in composition.
Physical property: Color, density, melting point, boiling point.
Chemical property: Reactivity, flammability, acidity.
Changes of Physical State
Matter changes state through processes such as melting, freezing, evaporation, condensation, and sublimation.
Melting: Solid to liquid
Freezing: Liquid to solid
Evaporation: Liquid to gas
Condensation: Gas to liquid
Sublimation: Solid to gas
Density Calculations
Density is a physical property defined as mass per unit volume. It is calculated using the formula:
Example: If a substance has a mass of 10 g and a volume of 2 mL, its density is .
Law of Conservation of Mass and Energy
The law of conservation of mass states that mass is neither created nor destroyed in a chemical reaction. The law of conservation of energy states that energy cannot be created or destroyed, only transformed.
Conservation of mass: Total mass of reactants equals total mass of products.
Conservation of energy: Energy changes form but total energy remains constant.
The Percent Concept
Percent is used to express the ratio of a part to the whole, multiplied by 100.
Example: If 20 g of salt is dissolved in 100 g of water, the percent salt is .
Ch.4 Models of the Atom
Atomic Notation and Subatomic Particles
Atoms are composed of protons, neutrons, and electrons. Atomic notation provides information about these particles.
Atomic number (Z): Number of protons in the nucleus.
Mass number (A): Sum of protons and neutrons.
Number of neutrons:
Number of electrons: Equal to protons in a neutral atom.
Isotopes and Average Atomic Weight
Isotopes are atoms of the same element with different numbers of neutrons. The average atomic weight is calculated using the masses and abundances of isotopes.
Example: If an element has two isotopes, A (mass 10, abundance 60%) and B (mass 12, abundance 40%), the average atomic weight is .
Electron Configuration
Electron configuration describes the arrangement of electrons in an atom. Electrons fill orbitals in order of increasing energy.
Example: The electron configuration for sodium (Na, atomic number 11) is 1s2 2s2 2p6 3s1.
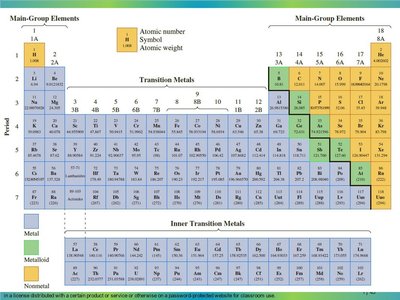
Periodic Table: Metals, Nonmetals, and Metalloids
The periodic table organizes elements by increasing atomic number and groups them by similar properties. Elements are classified as metals, nonmetals, or metalloids.
Metals: Good conductors, malleable, shiny (left and center of table).
Nonmetals: Poor conductors, brittle, dull (right side of table).
Metalloids: Properties intermediate between metals and nonmetals (border between metals and nonmetals).
Significant Figures and Scientific Notation
Significant figures reflect the precision of a measurement. Scientific notation expresses numbers as a product of a coefficient and a power of ten.
Significant figures: All nonzero digits, zeros between nonzero digits, and trailing zeros in the decimal portion are significant.
Scientific notation: , where and is an integer.
Example: 0.00456 =
Basic Calculations
Perform addition, subtraction, multiplication, and division using proper significant figures.
Addition/Subtraction: Result should have the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: Result should have the same number of significant figures as the measurement with the fewest significant figures.
Example Problems
Convert 250 milliliters to liters:
Calculate density: If mass = 15 g and volume = 3 mL,
Write electron configuration for carbon (atomic number 6): 1s2 2s2 2p2
