 Back
BackIonic and Molecular Compounds: Structure, Formation, and Naming
Study Guide - Smart Notes

Ionic and Molecular Compounds
Types of Compounds
Compounds are classified as either ionic or molecular (covalent) based on the nature of their constituent particles and the type of chemical bonds formed. Ionic compounds consist of ions held together by ionic bonds, while molecular compounds consist of molecules held together by covalent bonds.
Ionic Compounds: Composed of positive and negative ions (cations and anions) formed by the transfer of electrons.
Molecular Compounds: Composed of molecules formed by the sharing of electrons between nonmetals.
Examples: Na+ and Cl- ions (ionic), H2O and C2H6 molecules (molecular).
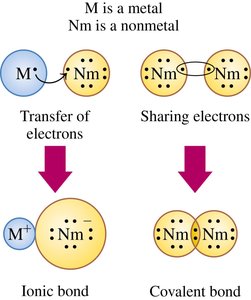
Formation of Ionic and Covalent Bonds
Electron Transfer and Sharing
The formation of chemical bonds involves either the transfer or sharing of electrons. Metals tend to lose electrons and form cations, while nonmetals gain electrons to form anions. Covalent bonds are formed when two nonmetals share electrons to achieve stable electron configurations.
Ionic Bond: Formed by the transfer of electrons from a metal to a nonmetal, resulting in oppositely charged ions.
Covalent Bond: Formed by the sharing of electrons between two nonmetals.
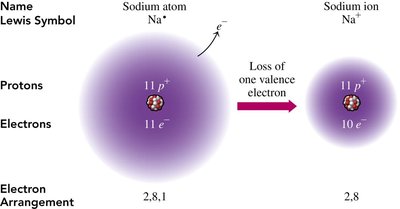
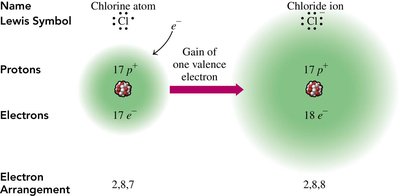
The Octet Rule and Ion Formation
Valence Electrons and Stability
Atoms tend to gain, lose, or share electrons to achieve a stable configuration with eight valence electrons, known as the octet rule. This rule explains the formation of ions and molecules.
Cations: Formed when metals lose electrons (e.g., Na+).
Anions: Formed when nonmetals gain electrons (e.g., Cl-).
Electron Arrangement: After ion formation, the electron configuration resembles that of a noble gas.
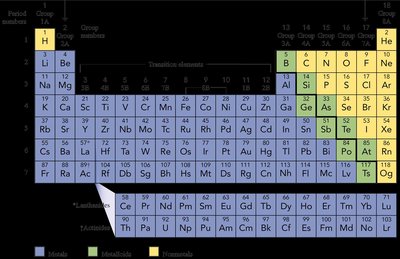
Periodic Table and Ion Formation
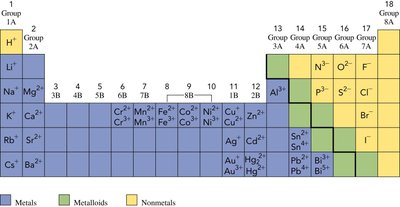
Group Trends and Ion Charges
The periodic table helps predict the charges of ions formed by elements. Metals in Groups 1A and 2A form cations with +1 and +2 charges, respectively. Nonmetals in Groups 5A, 6A, and 7A form anions with -3, -2, and -1 charges, respectively.
Group 1A: Forms +1 cations (e.g., Na+).
Group 2A: Forms +2 cations (e.g., Mg2+).
Group 7A: Forms -1 anions (e.g., Cl-).
Writing Formulas for Ionic Compounds
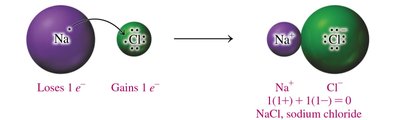
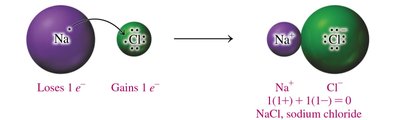
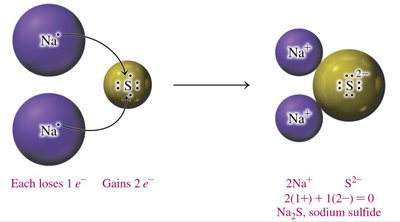
Charge Balance and Formula Units
Ionic compounds are written so that the total positive and negative charges balance to zero. The formula unit represents the lowest ratio of ions in the compound.
Charge Balance: The sum of cation and anion charges must equal zero.
Subscripts: Indicate the number of each ion needed for charge balance.
Cation First: The cation is written before the anion in the formula.
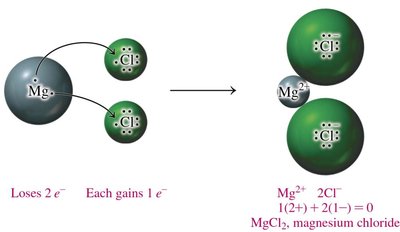
Naming Ionic Compounds
Monatomic and Polyatomic Ions
Ionic compounds are named by stating the cation first, followed by the anion. Monatomic ions are single atoms, while polyatomic ions are groups of atoms with an overall charge. Transition metals may have variable charges, indicated by Roman numerals.
Monatomic Ions: Named by element name (cation) or element root + "-ide" (anion).
Polyatomic Ions: Named by specific names (e.g., sulfate, nitrate).
Variable Charge Metals: Use Roman numerals to indicate charge (e.g., Iron(III) chloride).
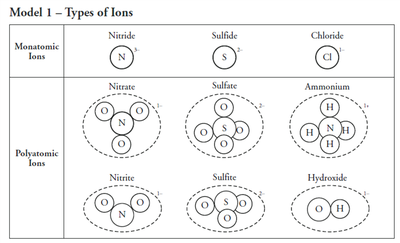
Polyatomic Ions
Structure and Naming
Polyatomic ions are groups of atoms covalently bonded, carrying an overall charge. Their names often end in "-ate" or "-ite" depending on the number of oxygen atoms. Some exceptions include ammonium (NH4+), hydroxide (OH-), and cyanide (CN-).
Charge: Usually -1, -2, or -3; ammonium is +1.
Association: Polyatomic ions combine with ions of opposite charge to form ionic compounds.
Examples: CaSO4 (plaster cast), NH4NO3 (fertilizer).
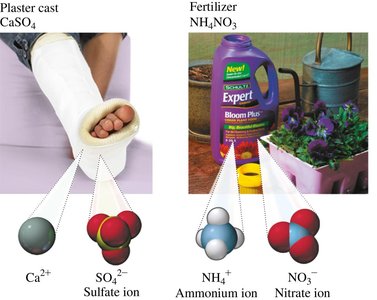
Molecular (Covalent) Compounds
Bond Formation and Naming
Molecular compounds are formed by the sharing of electrons between nonmetals. The number of shared electrons corresponds to the number needed to achieve a noble gas configuration. Naming uses prefixes to indicate the number of atoms.
Bond Formation: Number of bonds = 8 - (number of valence electrons).
Naming: First element uses its name; second element uses root + "-ide"; prefixes indicate quantity (mono-, di-, tri-, etc.).
Exceptions: Hydrogen (2 electrons), expanded octets for S, P, Cl, Br, I.
Electronegativity and Bond Polarity
Bond Types and Electron Distribution
Electronegativity is the ability of an atom to attract shared electrons. The difference in electronegativity determines whether a bond is ionic, polar covalent, or nonpolar covalent.
Ionic Bond: Large difference in electronegativity; electrons are transferred.
Polar Covalent Bond: Moderate difference; electrons are unequally shared.
Nonpolar Covalent Bond: Small or no difference; electrons are equally shared.
Attractive Forces in Compounds
Types of Intermolecular Forces
Particles in compounds experience attractive forces that influence their physical properties. Ionic compounds have strong electrostatic forces, while molecular compounds may have dipole-dipole, hydrogen bonding, or dispersion forces.
Ionic Compounds: Strong electrostatic attractions.
Polar Molecules: Dipole-dipole attractions and hydrogen bonding (when H is bonded to F, O, or N).
Nonpolar Molecules: Weak dispersion forces (temporary dipoles).
Melting Point: Stronger attractive forces result in higher melting points.
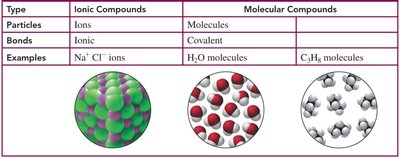
Summary Table: Ionic vs. Molecular Compounds
Type | Ionic Compounds | Molecular Compounds |
|---|---|---|
Particles | Ions | Molecules |
Bonds | Ionic | Covalent |
Examples | Na+, Cl- ions | H2O, C2H6 molecules |
Additional info: This summary table is based on the provided image and expands on the comparison of ionic and molecular compounds.
