 Back
BackLight, Quantum Mechanics, and Electron Configuration: Foundations of Atomic Structure
Study Guide - Smart Notes

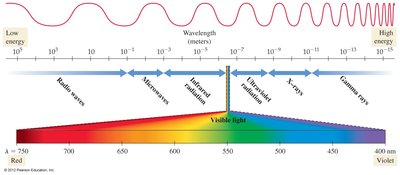
Nature of Light and the Electromagnetic Spectrum
What is Light?
Light is a form of electromagnetic radiation, a type of energy that travels through space at a constant speed of m/s. The electromagnetic spectrum encompasses all types of electromagnetic radiation, which differ in wavelength and frequency. Visible light is only a small portion of this spectrum, with wavelengths between approximately 400 nm (violet) and 750 nm (red).
Gamma rays: Shortest wavelength, highest energy; can damage biological molecules.
X-rays: Penetrate substances that block visible light; used in medical imaging.
Ultraviolet (UV): Causes sunburn; higher energy than visible light.
Visible light: Enables vision; photons rearrange molecules in the eye.
Infrared: Emitted by warm objects; felt as heat.
Microwaves: Longer wavelength, lower energy; used in communication and cooking.
Radio waves: Longest wavelength, lowest energy; used for radio, TV, and cell phones.
Color and Light
The color of an object is determined by the wavelengths of light it reflects. For example, a red shirt appears red because it reflects red light and absorbs other colors.

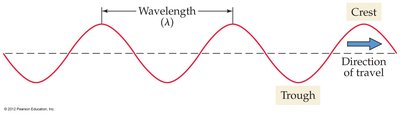
Waves: Wavelength and Frequency
Light behaves as a wave, characterized by its wavelength () and frequency ():
Wavelength (): The distance between adjacent wave crests.
Frequency (): The number of wave cycles passing a point per second (measured in Hz).
Wavelength and frequency are inversely related: , where is the speed of light.
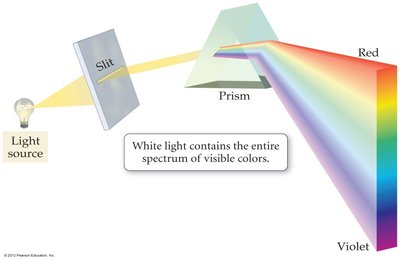
Separation of Light: The Prism
When white light passes through a prism, it separates into its constituent colors (spectrum), ranging from red (longest wavelength) to violet (shortest wavelength).
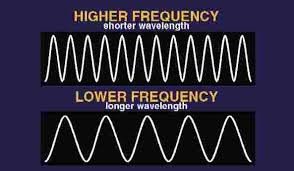
Relationship Between Wavelength and Frequency
Shorter wavelengths correspond to higher frequencies and higher energy per photon. Conversely, longer wavelengths have lower frequencies and lower energy.
Light as Particles: Photons and Energy
Photons
Light can also be described as a stream of particles called photons. The energy of a photon depends on its wavelength or frequency:
Energy per photon:
Where is Planck's constant ( J·s)
Violet light (shorter wavelength) carries more energy per photon than red light (longer wavelength).
Atomic Models: Bohr and Quantum Mechanics
The Bohr Model of the Atom
Niels Bohr proposed that electrons orbit the nucleus in fixed energy levels (quantized orbits). Each orbit is specified by a quantum number
Electrons cannot exist between orbits.
Energy is absorbed or emitted when an electron moves between orbits.
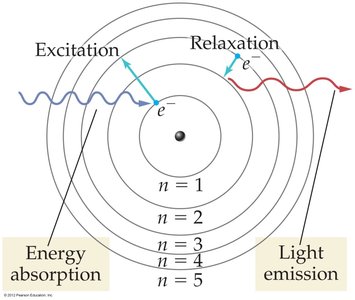
Excitation and Emission
When an atom absorbs energy, an electron is excited to a higher-energy orbit. When it relaxes back to a lower-energy orbit, it emits a photon of light with energy corresponding to the difference between the two orbits.
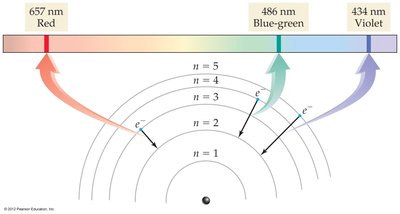
Hydrogen Emission Spectrum
The light emitted by excited hydrogen atoms consists of specific lines at specific wavelengths, each corresponding to a transition between two orbits. For example, the line at 486 nm corresponds to an electron moving from to .
Limitations of the Bohr Model
The Bohr model successfully explained the hydrogen spectrum but failed for atoms with more than one electron. It was replaced by the quantum-mechanical model, which uses probability maps (orbitals) to describe electron locations.
The Quantum-Mechanical Model of the Atom
Orbitals and Quantum Numbers
In the quantum-mechanical model, electrons occupy orbitals, which are three-dimensional probability maps. The principal quantum number () describes the size and energy of the orbital, while the angular momentum quantum number () describes its shape:
s orbital: Spherical shape ()
p orbital: Dumbbell shape ()
d orbital: Cloverleaf shape ()
f orbital: Complex shape ()
Shell | Number of subshells | Letters specifying subshells |
|---|---|---|
n = 4 | 4 | s, p, d, f |
n = 3 | 3 | s, p, d |
n = 2 | 2 | s, p |
n = 1 | 1 | s |
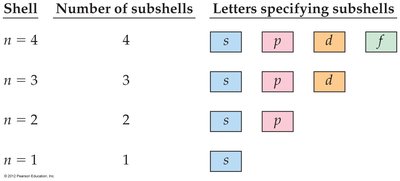
Principal Shells and Subshells
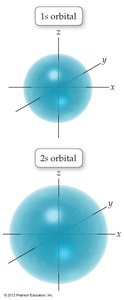
The number of subshells in a principal shell equals the value of . For example, has two subshells: 2s and 2p. The 2s orbital is similar to the 1s orbital but larger in size.
Ground and Excited States
The ground state is the lowest energy state of an atom, with electrons in the lowest possible orbitals. When energy is absorbed, electrons can move to higher-energy (excited) states.
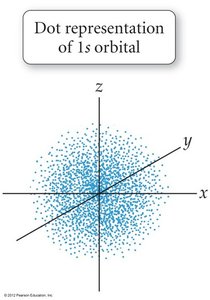
Representations of Orbitals
Orbitals are often represented by dots, where dot density indicates the probability of finding an electron. The 1s orbital is most dense near the nucleus.

Energy Ordering of Orbitals
In multi-electron atoms, subshells within a principal shell have different energies due to electron–electron interactions. The order of filling is not strictly by ; for example, 4s fills before 3d.
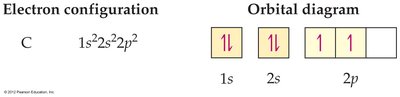
Electron Configurations and Orbital Diagrams
Electron Configuration
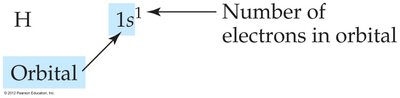
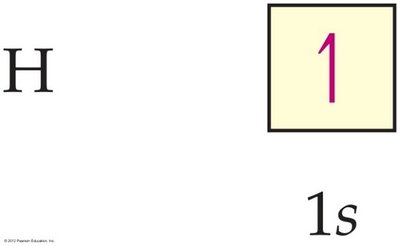
An electron configuration shows the occupation of orbitals by electrons. For hydrogen, the configuration is 1s1.
Orbital Diagrams
Orbital diagrams use boxes to represent orbitals and arrows for electrons. The direction of the arrow indicates electron spin. The Pauli exclusion principle states that an orbital can hold no more than two electrons, which must have opposite spins.
Hund's Rule
When filling orbitals of equal energy (degenerate orbitals), electrons fill them singly first, with parallel spins, before pairing up. This is known as Hund's rule.
Examples of Electron Configurations
Helium (He): 1s2
Lithium (Li): 1s22s1
Carbon (C): 1s22s22p2



Order of Orbital Filling
Orbitals fill in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, etc. Lower-energy orbitals fill before higher-energy orbitals.

Noble Gas Core Notation
For elements beyond neon, electron configurations can be abbreviated using the previous noble gas in brackets. For example, sodium (Na): [Ne]3s1.
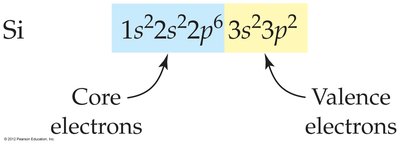
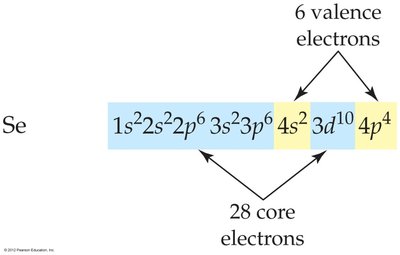
Valence and Core Electrons
Valence electrons are those in the outermost principal shell and are involved in chemical bonding. Core electrons are all other electrons.
Periodic Trends and the Quantum Model
Periodic Trends in Electron Configurations
The number of valence electrons for main-group elements equals the group number. The row number equals the highest principal shell ( value). Transition metals have unique trends, with the d orbital's principal quantum number equal to the row number minus one.
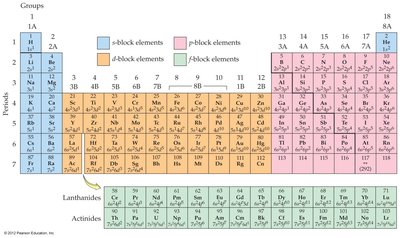
Stability of Noble Gases
Atoms with 8 valence electrons (or 2 for helium) are particularly stable (noble gases). Elements close to noble gas configurations are highly reactive, as they tend to gain or lose electrons to achieve stability.

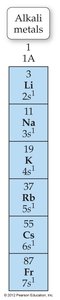
Alkali and Alkaline Earth Metals
Alkali metals (Group 1) have one valence electron and tend to form 1+ cations. Alkaline earth metals (Group 2) have two valence electrons and tend to form 2+ cations.

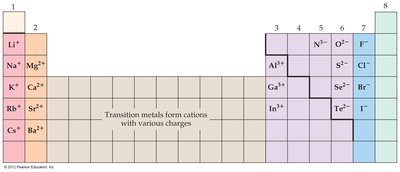
Formation of Predictable Ions
Main-group elements form predictable ions to achieve noble gas configurations. Transition metals can form various cations.
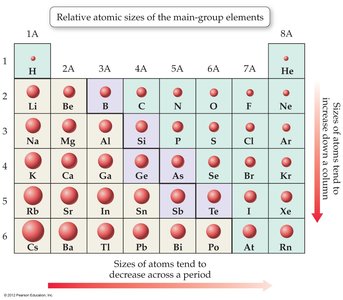
Periodic Trends: Atomic Size
Atomic size decreases across a period (left to right) due to increasing nuclear charge, which pulls electrons closer. Atomic size increases down a group due to additional electron shells.
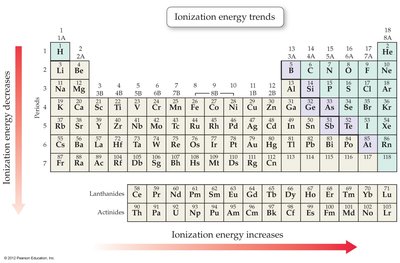
Periodic Trends: Ionization Energy
Ionization energy is the energy required to remove an electron from a neutral atom. It increases across a period and decreases down a group.
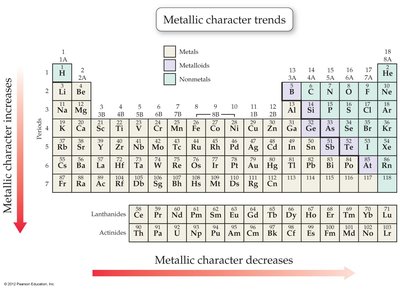
Periodic Trends: Metallic Character
Metals tend to lose electrons, while nonmetals tend to gain them. Metallic character decreases across a period and increases down a group.
Additional info: The quantum-mechanical model provides the foundation for understanding chemical bonding, periodic trends, and the reactivity of elements. Mastery of electron configurations and periodic trends is essential for predicting chemical behavior.
