 Back
BackLiquids, Solids, and Intermolecular Forces: Study Guide
Study Guide - Smart Notes

Liquids, Solids, and Intermolecular Forces
Intermolecular Forces: Concepts and Types
Intermolecular forces are the attractive forces that occur between molecules and play a crucial role in determining the physical properties of substances. They are distinct from intramolecular forces, which are the chemical bonds within a molecule.
Intramolecular Forces: Exist within a molecule, bond atoms together, and influence chemical properties. Examples include ionic and covalent bonds. These are stronger than intermolecular forces.
Intermolecular Forces: Exist between molecules and influence physical properties such as boiling point, melting point, and solubility. They hold liquid and solid molecules together.
Key Point: The dominant forces between molecules (intermolecular forces) are electrostatic in origin.
Intermolecular forces are weaker than chemical bonds.
They are responsible for the physical state (solid, liquid, gas) and properties of substances.
Types of Intermolecular Forces
There are four main types of intermolecular forces that hold molecules together. The polarity of compounds plays a significant role in determining the type of force present.
London Dispersion Forces (van der Waals): Present in all molecules, including nonpolar compounds. Strength increases with molecular mass.
Dipole-Dipole Forces: Occur between two polar covalent compounds.
Hydrogen Bonding: Occurs in compounds containing hydrogen directly bonded to F, O, or N.
Ion-Dipole Forces: Occur between ions and polar compounds. Example: NaCl & H2O.
Example: The major type of intermolecular force between N2 is London dispersion; between CH3OH is hydrogen bonding; between CH3Cl is dipole-dipole; between KCl & CH3OH is ion-dipole.
Intermolecular Forces and Physical Properties
Intermolecular forces directly influence measurable physical properties such as boiling point, melting point, vapor pressure, viscosity, and surface tension.
Direct Relationships: Stronger intermolecular forces lead to higher boiling points, melting points, and surface tension.
Indirect Relationships: Stronger intermolecular forces lead to lower vapor pressure and lower volatility.
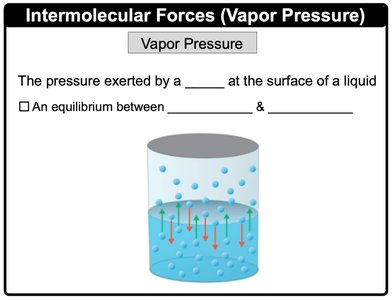
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor at the surface of a liquid. It represents an equilibrium between evaporation and condensation.
Boiling Point, Melting Point, Surface Tension, and Viscosity
Boiling Point (BP): Temperature where vapor and liquid are in equilibrium.
Melting Point (MP): Temperature where solid and liquid are in equilibrium.
Surface Tension: Measure of intermolecular forces on a liquid surface.
Viscosity: Resistance to flow; higher viscosity means slower movement. Temperature increases generally decrease viscosity.

Classification of Solids
Solids are classified based on the arrangement of their particles and the nature of their bonding.
Crystalline Solids: Atoms, ions, or molecules are arranged in a highly ordered, repeating pattern.
Amorphous Solids: Particles are randomly arranged with no discernible pattern.
Types of Solids
Ionic Solids: Composed of ions held together by electrostatic attraction. Example: NaCl.
Molecular Solids: Composed of molecules held together by intermolecular forces. Example: CO2.
Covalent Network Solids: Atoms connected by covalent bonds in a network. Example: graphite.
Metallic Solids: Metal atoms held together by metallic bonds (pooling of electrons).
Amorphous Solids: No regular arrangement; examples include plastics and glass.
Heating and Cooling Curves
Heating and cooling curves represent the amount of heat absorbed or released during phase changes. They show the relationship between temperature and time as a substance changes state.
Specific Heat: The amount of heat required to raise the temperature of 1 gram of a substance by 1°C.
Heat of Fusion (ΔHfusion): The energy required to convert a solid to a liquid at its melting point.
Heat of Vaporization (ΔHvaporization): The energy required to convert a liquid to a gas at its boiling point.
Example Calculation: To calculate the energy required for a phase change, use:
For temperature change:
For phase change:
Example: How much energy is required to convert 76.4 g acetone from liquid at -30°C to solid at -115°C? Use specific heat and heat of fusion values as needed.
Additional info: Heating and cooling curves are essential for understanding phase transitions and energy calculations in chemistry.
