 Back
BackMatter and Energy: States, Properties, Changes, and Energy in Chemistry
Study Guide - Smart Notes

Matter & Its States
States of Matter
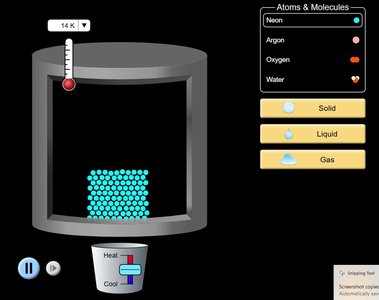
The three primary states of matter are solid, liquid, and gas. Each state is characterized by the arrangement and movement of its particles, as well as its volume and shape properties.
Solids: Particles are closely packed in an ordered structure. Solids have a definite shape and volume.
Liquids: Particles are close but not ordered. Liquids have a definite volume but a variable shape.
Gases: Particles are far apart and not ordered. Gases have variable shape and volume.
Definite means fixed or unchanging, while variable means able to change.
Classifying Matter
Types of Matter
Matter can be classified as pure substances or mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Element: A substance made of only one type of atom.
Compound: A substance made of two or more types of atoms chemically bonded.
Homogeneous mixture: Uniform composition throughout (e.g., salt water).
Heterogeneous mixture: Non-uniform composition (e.g., salad).
Physical & Chemical Properties
Properties of Gold
Physical properties can be measured or observed without changing the identity of the substance. Chemical properties can only be observed during a change in the substance's identity.
Physical Properties: Color, density, melting point, conductivity.
Chemical Properties: Flammability, ability to react with oxygen.
Physical properties are consistent between samples of the same substance, while chemical properties are revealed only during chemical changes.

Physical & Chemical Changes
Changes in Matter
A physical change alters the form or appearance of matter but does not change its identity. A chemical change (chemical reaction) alters the identity of the particles, forming new substances.
Physical Change: Cutting, melting, boiling.
Chemical Change: Burning, rusting, decomposition.
Physical changes may be reversible or irreversible, but the substance remains the same. Chemical changes can also be reversible or irreversible, but new substances are formed.


Chemical Changes
Signs of Chemical Reactions
Chemical changes are often accompanied by observable signs:
Color change
Production of gas
Heat or light emission
Formation of a precipitate
In a chemical reaction, reactants are transformed into products.


Conservation of Mass
Law of Conservation of Mass
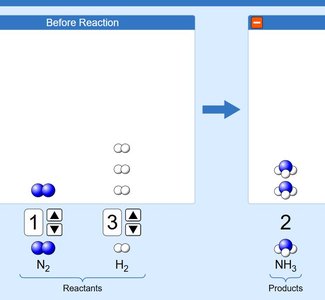
In a chemical reaction, the total mass of the reactants equals the total mass of the products. Atoms are rearranged, but their number and identity remain unchanged.
Mass is not created or destroyed in ordinary chemical reactions.
Balancing chemical equations ensures conservation of mass.
Example: Burning 12.0 g of carbon in 32.0 g of oxygen produces 44.0 g of carbon dioxide.

Conservation of Energy
What is Energy?
Energy is the ability to do work or cause change. In chemistry, energy is involved in moving particles and driving reactions.
Kinetic Energy: Energy of movement.
Potential Energy: Stored energy.
Thermal Energy: Energy related to heat.
Chemical Energy: Energy stored in chemical bonds.
Energy is not created or destroyed, only transformed from one form to another.


Tracking & Transforming Energy
Chemical Reactions and Energy
When fuels like methane are burned, chemical energy is released as heat. The chemical equation for burning methane is:
No atoms are lost; they are rearranged.
Energy is transformed, not lost.


Energy Units
Measuring Energy
Energy is measured in joules (J) and calories (cal). Joules are used in physics, while calories are tied to heat. Food labels use kilocalories (kcal).
1 calorie = 4.18 joules

Tracking Energy
System and Surroundings
To track energy changes, chemists define the system (the part of the universe being studied) and the surroundings (everything else).
Energy changes are tracked for the system.
Example: An apple is the system; the tree, air, and ground are the surroundings.


Endothermic & Exothermic Processes
Heat Flow in Chemistry
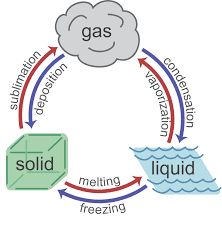
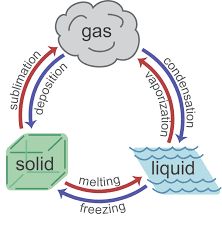
Endothermic processes absorb heat, while exothermic processes release heat. These terms are used to describe both chemical and physical changes.
Endothermic: Heat flows into the system (e.g., melting, vaporization).
Exothermic: Heat flows out of the system (e.g., freezing, condensation).

Heat vs Temperature
Definitions and Units
Heat is the flow of energy from a warmer object to a cooler one. Temperature is a measure of the average kinetic energy of particles. Common units for temperature are Celsius (°C), Fahrenheit (°F), and Kelvin (K).

Temperature Units Interlude
Reference Points and Conversions
The three temperature scales have different zero points:
Celsius: 0°C is the freezing point of pure water.
Fahrenheit: 0°F is the coldest salty water as a liquid.
Kelvin: 0 K is absolute zero, the lack of molecular motion.
Celsius and Kelvin degrees are the same size. Most chemistry calculations use Kelvin.


Conversion formula:
Temperature vs Heat
Comparing Heat and Temperature
Two objects at the same temperature can have different amounts of heat energy, depending on their mass and substance.
Heat energy depends on mass, temperature, and substance identity.
Heat flows when there is a temperature difference.
Specific Heat Capacity
Definition and Calculation
Specific heat capacity is the amount of energy required to raise the temperature of one gram of a substance by one degree Celsius. It is represented by "s" in the equation:
q: Heat energy (Joules)
m: Mass (grams)
ΔT: Change in temperature (final - initial)
Example: Cooling 225 g of water from 60°C to 20°C. Calculate q using the specific heat capacity of water.
Specific heat capacity varies between substances; water has a high specific heat, copper has a low specific heat.
Heat flows from the system to the surroundings in exothermic processes.
For cooling water:
For cooling copper:
Practical implication: Copper warms up the air less than water for the same mass and temperature change.
