 Back
BackMeasurement and Problem Solving in Chemistry: Units, Significant Figures, and Dimensional Analysis
Study Guide - Smart Notes

Chapter 2: Measurement and Problem Solving
Introduction to Measurement in Chemistry
Chemistry relies on precise and accurate measurements to describe matter and its changes. Understanding measurement is essential for quantifying mass, volume, temperature, and time, which are foundational to experimental science.
Mass and Volume: Quantify the amount of substance present.
Temperature: Indicates thermal energy and reaction conditions.
Time: Measures the duration of chemical processes.
Units: Specify what is being measured and ensure clarity in communication.
Scientific Notation
Scientific notation is used to express very large or very small numbers in a compact form, making calculations easier and reducing errors.
Format: where A is the decimal part and n is the exponent.
Example: (Avogadro's number)
Decimal Part (A): Must be between 1 and 10 (e.g., 1.22, 3.4, 5.67).
Exponent (n): Indicates the number of places the decimal moves.
Uncertainty in Measurements
All measurements have inherent uncertainty. The last digit in any measurement is always estimated, reflecting the precision of the measuring device.
Estimated Digit: Always include one more digit beyond the device's precision.
Significance: The extra digit increases accuracy in calculations.
Exact Numbers: If a measurement is exact, extra zeros may be used (e.g., 24.0 mm).
Significant Figures (Sig Figs)
Significant figures indicate the precision of a measurement. They include all known digits plus one estimated digit.
Non-zero digits: Always significant (e.g., 4.567 has 4 sig figs).
Sandwiched zeros: Significant if between non-zero digits (e.g., 1,300.9 has 5 sig figs).
Trailing zeros: Significant if a decimal is present (e.g., 14.900000 has 8 sig figs).
Leading zeros: Not significant; they are placeholders (e.g., 0.000045 has 2 sig figs).
Exact numbers: Have infinite significant figures (e.g., 12 in a dozen).
Counted items: Fixed numbers, not estimates (e.g., 5 fingers).
Scientific Notation and Sig Figs: The exponent does not affect the number of significant figures; only the decimal part matters.
Example: 1200 (2 SF), 1.20 x 10^3 (3 SF), 1.200 x 10^3 (4 SF)
Significant Figures in Calculations
When performing calculations, the precision of the result is limited by the least precise measurement.
Multiplication/Division: Round to the fewest significant figures in any number used.
Addition/Subtraction: Round to the fewest decimal places present in any number used.
Examples:
2.6 mm (2 SF) vs. 2.53 mm (3 SF) vs. 2.5217 mm (5 SF)
Multiplication: (2 SF)
Addition: (1 decimal place)
Rounding Rules for Significant Figures
Proper rounding ensures that results reflect the correct level of precision.
Round down: If the digit dropped is 4 or less.
Round up: If the digit dropped is 5 or more.
Special case: Rounding up a 9 may carry over to the next digit (e.g., 0.09995 to 0.100).
Dimensional Analysis and Conversion Factors
Dimensional analysis uses conversion factors to solve problems involving different units. Conversion factors are ratios that express the same quantity in different units.
Example:
Example:
Area:
Volume:
Density:
Basic Units of Measurement
Understanding the basic and derived units is essential for accurate measurement and calculation in chemistry.
Length: meter (m), centimeter (cm), millimeter (mm), inch (in), foot (ft), mile (mi)
Mass: kilogram (kg), gram (g), pound (lb), ounce (oz)
Volume: liter (L), milliliter (mL), cubic centimeter (cm3), quart (qt), gallon (gal)
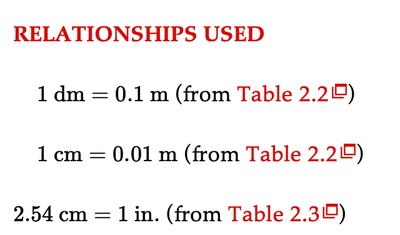
Relationships Used in Conversions
Key relationships are used to convert between units in dimensional analysis.
Density and Its Applications
Density is a derived unit that relates mass to volume. It is useful for identifying substances and solving problems involving mass and volume.
Formula:
Units: g/cm3, g/mL, g/L
Example:
Comparison: Substances have characteristic densities (e.g., water = 1.0 g/cm3, gold = 19.3 g/cm3).
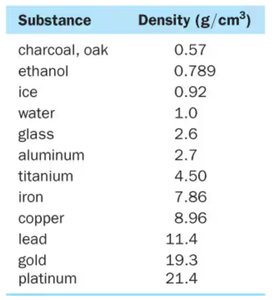
Substance | Density (g/cm3) |
|---|---|
Charcoal, oak | 0.57 |
Ethanol | 0.789 |
Ice | 0.92 |
Water | 1.0 |
Glass | 2.6 |
Aluminum | 2.7 |
Titanium | 4.50 |
Iron | 7.86 |
Copper | 8.96 |
Lead | 11.4 |
Gold | 19.3 |
Platinum | 21.4 |
Summary Table: Significant Figures Rules
Rule | Example | Sig Figs |
|---|---|---|
Non-zero digits | 4.567 | 4 |
Sandwiched zeros | 1,300.9 | 5 |
Trailing zeros (with decimal) | 14.900000 | 8 |
Trailing zeros (no decimal) | 13,000 | 2 |
Leading zeros | 0.000045 | 2 |
Exact numbers | 12 eggs | Infinite |
Additional info:
Dimensional analysis is a powerful tool for solving word problems and ensuring correct units in calculations.
Always include units in answers to maintain clarity and accuracy.
Multi-step problems require careful tracking of significant figures at each stage; only round at the final step.
