 Back
BackMeasurement and Problem Solving in Introductory Chemistry
Study Guide - Smart Notes

Measurement and Problem Solving
Introduction to Measurement in Chemistry
Measurement is a foundational aspect of chemistry, allowing scientists to quantify observations and communicate results precisely. Understanding how to measure, record, and interpret data is essential for success in laboratory and theoretical chemistry.
Scientific Notation
Purpose and Use of Scientific Notation
Scientific notation is a method used to express very large or very small numbers in a concise form. This is especially important in chemistry, where measurements can span many orders of magnitude.
Definition: Scientific notation expresses numbers as a product of a coefficient (between 1 and 10) and a power of ten.
Purpose: Makes calculations and comparisons easier and reduces errors when handling extreme values.
Example: meters is written as meters.
Order of Magnitude: The exponent in scientific notation indicates the order of magnitude, which helps compare the relative size of numbers.
Steps for Writing Numbers in Scientific Notation
Write the first non-zero digit, followed by a decimal point.
Copy the remaining significant digits.
Add “”.
Count how many places the decimal moved to determine the exponent.
If the original number is greater than 1, the exponent is positive; if less than 1, it is negative.
Example: L is L.
Comparing Numbers in Scientific Notation
To compare, convert both numbers to standard notation or compare exponents directly.
Zeroes written explicitly in scientific notation are significant and affect precision.
Calculator Usage for Scientific Notation
Most scientific calculators have an “EE” or “EXP” button to enter numbers in scientific notation efficiently.

Significant Figures
Definition and Importance
Significant figures (sig figs) indicate the precision of a measured or calculated quantity. They reflect all known digits plus one estimated digit, showing the reliability of the measurement.
Example: 700, 700., 700.0, and 700.00 all have different numbers of significant figures, indicating different levels of precision.
Rules for Counting Significant Figures
Rule 1: All non-zero digits are significant.
Rule 2: Zeros between non-zero digits are significant.
Rule 3: Leading zeros are not significant.
Rule 4: Trailing zeros are significant only if there is a decimal point.
Rule 5: Exact numbers (from counting, not measurement) have infinite significant figures.
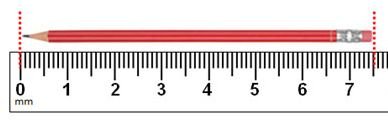
Reading Measurement Tools
When using a ruler or other measuring device, always estimate one digit beyond the smallest marked unit. This estimated digit is also significant.
Significant Figures in Calculations
Addition/Subtraction: The result should have the same number of decimal places as the measurement with the fewest decimal places.
Multiplication/Division: The result should have the same number of significant figures as the measurement with the fewest significant figures.
Example: (should be rounded to 8.1 cm2 if both have two sig figs).
Combined Calculations
When calculations involve both addition/subtraction and multiplication/division, follow the rules for each operation in sequence, rounding at the end.
SI (Metric) Units and Conversions
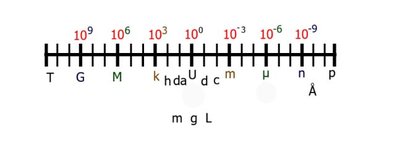
Base Units and Prefixes
The metric system uses base units for length (meter), mass (gram), and volume (liter). Prefixes indicate multiples or fractions of these units, making conversions straightforward.
Meter (m): Measures distance
Liter (L): Measures volume
Gram (g): Measures mass
Metric Conversions
Move the decimal point according to the metric prefix.
For more complex conversions, convert to the base unit first, then to the desired unit.
Example:
Dimensional Analysis and Conversion Factors
General Approach
Dimensional analysis is a systematic method for converting between units using conversion factors. It ensures that units cancel appropriately, leading to the correct answer.
Write the given value with its unit.
Multiply by conversion factors so that units cancel.
Continue until the desired unit is reached.
Round the final answer according to significant figures.
Example: To convert 6.50 cm to feet, use the conversion factors and .
Exact vs. Approximate Conversion Factors
Some conversion factors are defined and considered infinitely precise (e.g., ).
Others are measured and have limited precision (e.g., ).
For this course, treat all conversion factors as infinitely precise unless otherwise noted.
Complex Units
When converting units with numerators and denominators (e.g., m/s to mi/hr), treat each part independently and ensure all units cancel appropriately.
Density
Definition and Calculation
Density is a measure of how much mass is contained in a given volume. It is a key property for identifying substances and solving various chemistry problems.
Formula:
Common units: g/mL or g/cm3
Example: Water has a density of .
Density can also be used as a conversion factor to relate mass and volume.
Applications of Density
Determining whether an object will sink or float (compare its density to that of water).
Calculating the mass or volume of a substance given its density and one other property.
Example Problem: If platinum has a density of and a ring has a mass of , the volume of water displaced is .
