 Back
BackMeasurement and Problem Solving: Scientific Notation, Significant Figures, and Units
Study Guide - Smart Notes

Measurement and Problem Solving
Scientific Notation
Scientific notation is a method used to express very large or very small numbers in a concise format, making calculations easier and reducing errors. It is commonly used in chemistry to represent quantities such as atomic sizes or astronomical distances.
Format: A number is written as the product of a coefficient (between 1 and 10) and a power of ten. For example, 94,500,000 miles becomes miles.
Examples:
Distance from Earth to Sun: miles = miles
Size of a hydrogen atom: meters = meters
Conversion Practice: Numbers such as 582, 0.076, 4,700.0, and 25,800,000.0 should be written in scientific notation without changing the number of digits.
Important Note: Never place a dash (-) in front of a number unless indicating a negative value.
Measurement and Uncertainty
Measurements in chemistry are fundamental to experimentation and problem solving. Every measurement consists of two parts: a numerical value and a unit. Measurements are never exact and always have some degree of uncertainty.
Numerical Value: Indicates the magnitude of the measurement.
Unit: Specifies what is being measured (e.g., kilograms, meters).
Uncertainty: All measurements have uncertainty due to limitations in the measuring instrument and estimation.
Precision: The place of uncertainty indicates the precision of the measurement.
Example: Is 8 inches equal to 8.0 inches? No, because 8.0 inches is more precise.
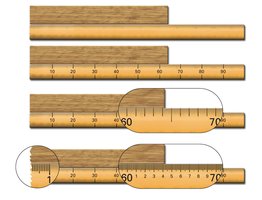
Recording Measurements
When recording a measurement, include all digits known with certainty plus one digit that is estimated. These digits are called significant figures.
Significant Figures: Digits in a measured number that are known for certain, plus one estimated digit.
Example: If an object extends past 60 cm but not quite to 70 cm, estimate the ones place for the uncertain digit.
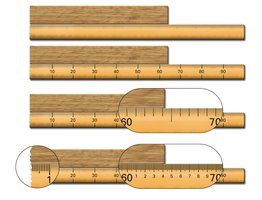
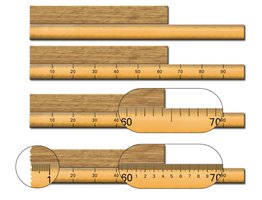
Significant Figures: Rules
Significant figures are essential for expressing the precision of measurements. The following rules help identify significant figures in a number:
All nonzero digits are significant.
Interior zeros (between two numbers) are significant.
Trailing zeros after a decimal point are significant.
Trailing zeros before a decimal point are significant.
Leading zeros (before the first nonzero digit) are not significant.
Trailing zeros at the end of a number, but before an implied decimal point, are ambiguous and should be avoided.
Identifying Exact Numbers
Exact numbers have an unlimited number of significant figures. They include:
Counting discrete objects (e.g., 10 pencils).
Integral numbers in equations (e.g., radius = diameter/2).
Defined quantities (e.g., 1 in = 2.54 cm).
Significant Figures in Scientific Notation
When converting a number to scientific notation, the number of significant figures remains unchanged.
Example: has 4 significant figures.
Example: has 3 significant figures.
Significant Figures in Calculations
When performing calculations, the result must reflect the precision of the data. Rounding rules and significant figure rules differ for multiplication/division and addition/subtraction.
Rounding: Round only the final answer, not intermediate steps.
Multiplication/Division: The result has the same number of significant figures as the factor with the fewest significant figures.
Addition/Subtraction: The result has the same number of decimal places as the quantity with the fewest decimal places.
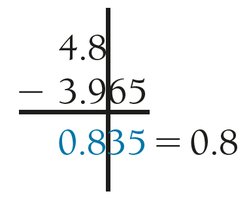
Addition/Subtraction with Scientific Notation
When adding or subtracting numbers in scientific notation:
Align exponents (rewrite numbers to share the same power of 10).
Add or subtract the decimal numbers as usual.
Identify the least precise number (fewest decimal places).
Round the result to match the decimal place of the least precise number.
Verify the final answer is in proper scientific notation.
Complex Calculations: Order of Operations
For calculations involving both multiplication/division and addition/subtraction, follow the standard order of operations (PEMDAS): Parentheses, Exponents, Multiplication/Division, Addition/Subtraction. Determine significant figures for intermediate answers without rounding until the final step.
Logs and Antilogs
When taking logarithms or antilogarithms:
Logarithms: The number of significant figures in the original number determines the number of decimal places in the result.
Antilogs: The number of decimal places in the original number determines the number of significant figures in the result.
Example: (4 significant figures) yields a result with 4 decimal places: -8.4887.
Example: (2 decimal places) yields a result with 2 significant figures: .
The Basic Units of Measurement
International System of Units (SI)
The SI system is the standard for scientific measurements. It is based on the metric system and includes base units for length, mass, volume, and other quantities.
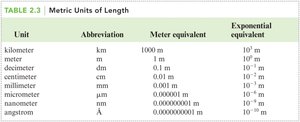
Metric Units of Length
The metric system uses multiples of ten for easy conversion between units. Common units include meter (m), centimeter (cm), millimeter (mm), and kilometer (km).
Unit | Abbreviation | Meter equivalent | Exponential equivalent |
|---|---|---|---|
Kilometer | km | 1000 m | m |
Meter | m | 1 m | m |
Decimeter | dm | 0.1 m | m |
Centimeter | cm | 0.01 m | m |
Millimeter | mm | 0.001 m | m |
Micrometer | μm | 0.000001 m | m |
Nanometer | nm | 0.000000001 m | m |
Angstrom | Å | 0.0000000001 m | m |
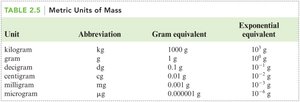
Metric Units of Mass
Unit | Abbreviation | Gram equivalent | Exponential equivalent |
|---|---|---|---|
Kilogram | kg | 1000 g | g |
Gram | g | 1 g | g |
Decigram | dg | 0.1 g | g |
Centigram | cg | 0.01 g | g |
Milligram | mg | 0.001 g | g |
Microgram | μg | 0.000001 g | g |
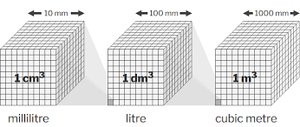
Metric Units of Volume
The SI unit for volume is the cubic meter (), but for liquids and gases, the liter (L) is more commonly used.
1 L = 1000 cm3 = 1 dm3
1 mL = 1 cm3
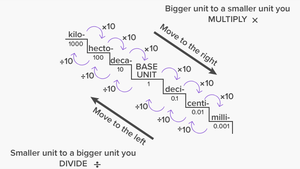
Metric Conversions
Conversions between metric units are based on powers of ten. Moving from a larger unit to a smaller unit, multiply; from a smaller unit to a larger unit, divide.
Problem Solving and Dimensional Analysis
Unit Conversion Problems
Unit conversion is a fundamental skill in chemistry. Conversion factors are used to relate different units and solve problems such as "How many days in 3 weeks?" or "How many inches in 4 feet?"
Conversion Factors: Equalities such as 2.54 cm = 1 in, 12 in = 1 foot, 5280 ft = 1 mile.
Dimensional Analysis: Use conversion factors to cancel units and solve for the desired quantity.
Example: To convert the length of a marathon (26.218 miles) to meters, use the appropriate conversion factors.
Density
Definition and Calculation
Density is a physical property defined as mass per unit volume. It is temperature dependent because volume changes with temperature.
Formula:
Example: If a metal has a mass of 427 g and a volume of 35.0 mL, its density is .
Density as a Conversion Factor: The density of gold is 19.3 g/cm3. A 25.0 cm3 volume of gold has a mass of g.
Additional info: Density is often used to identify substances and to convert between mass and volume in laboratory calculations.
