 Back
BackMeasurement and Problem Solving: Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Measurement and Problem Solving
Scientific Notation
Scientific notation is a method used to express very large or very small numbers in a concise form. It consists of two parts: a decimal part (between 1 and 10) and an exponential part (10 raised to an exponent).
Decimal Part: A number between 1 and 10.
Exponential Part: 10n, where n is an integer.
Positive Exponent: Indicates multiplication by 10 n times.
Negative Exponent: Indicates division by 10 n times.
Conversion Steps: Move the decimal point to obtain a number between 1 and 10, then multiply by 10 raised to the appropriate power.
Direction of Decimal Movement: Left movement yields a positive exponent; right movement yields a negative exponent.
Uncertainty in Measurement
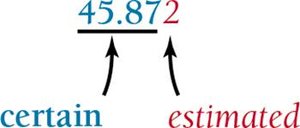
All measurements in science carry some degree of uncertainty. The uncertainty is reflected in the last digit reported, which is estimated. The more digits reported, the greater the precision.
Reporting: The last digit is uncertain; all others are certain.
Example: Reporting a temperature increase of 0.6°C means the true value could be between 0.5°C and 0.7°C.

Significant Figures
Significant figures indicate the precision of a measurement. The rules for determining significant figures are as follows:
All nonzero digits are significant.
Interior zeros (between nonzero digits) are significant.
Trailing zeros after a decimal point are significant.
Trailing zeros before a decimal point are significant.
Leading zeros (before the first nonzero digit) are not significant.
Trailing zeros before an implied decimal point are ambiguous.
Exact Numbers
Exact counting: Unlimited significant figures (e.g., 10 pencils).
Integral numbers in equations: Unlimited significant figures (e.g., radius = diameter/2).
Defined quantities: Unlimited significant figures (e.g., 1 in = 2.54 cm).
Estimating Measurements
When reading scales, estimate one digit beyond the smallest marking.
Estimating tenths: If the scale is marked every 1 gram, estimate to the tenths place.
Estimating hundredths: If the scale is marked every 0.1 gram, estimate to the hundredths place.


Significant Figures in Calculations
When performing calculations, the number of significant figures in the result depends on the operation:
Multiplication/Division: The result has the same number of significant figures as the factor with the fewest significant figures.
Addition/Subtraction: The result has the same number of decimal places as the quantity with the fewest decimal places.
Rounding: Round only the final answer, not intermediate steps. If the last digit dropped is 4 or less, round down; if 5 or more, round up.
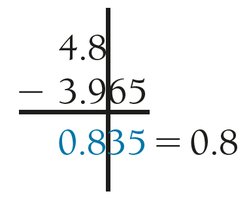
Combined Calculations
For calculations involving both multiplication/division and addition/subtraction, follow the order of operations and apply the appropriate significant figure rules at each step. Do not round intermediate results; underline the least significant figure as a reminder.
Basic Units of Measurement
The International System of Units (SI) is used in science. The main SI base units are:
Length: Meter (m) – defined as the distance light travels in a vacuum in 1/299,792,458 seconds.
Mass: Kilogram (kg) – defined using Planck’s constant.
Time: Second (s) – defined by the frequency of radiation from cesium-133 atoms.
Weight vs. Mass
Mass: Quantity of matter in an object; does not depend on gravity.
Weight: Gravitational pull on an object; depends on gravity.
SI Prefix Multipliers
Prefix multipliers are used to express measurements conveniently. Choose a prefix that matches the size of the quantity measured (e.g., picometer for chemical bonds).
Volume as a Derived Unit
Volume is a derived unit, calculated by cubing a unit of length. Common units include cubic meters (m3), cubic centimeters (cm3), and cubic millimeters (mm3).
Problem-Solving and Unit Conversions
Many chemistry problems involve converting units. Dimensional analysis is used to ensure units are handled correctly throughout calculations.
Always write numbers with units.
Units are multiplied, divided, and canceled like algebraic quantities.
Conversion factors: Constructed from equivalent quantities (e.g., 1 in = 2.54 cm).
Conversion factors can be inverted.
Solution Maps
A solution map visually outlines the steps required to solve a unit conversion problem.
General Problem-Solving Strategy
Sort: Organize the information given.
Strategize: Create a solution map.
Solve: Perform calculations, paying attention to significant figures.
Check: Ensure the answer makes physical sense and units are correct.
Solving Multistep Unit Conversion Problems
Each step in a multistep conversion uses a conversion factor with the previous unit in the denominator and the next unit in the numerator. The answer is rounded to the correct number of significant figures.
Unit Conversion in Both Numerator and Denominator
Some problems require converting units in both the numerator and denominator (e.g., mi/gal to km/L).
Converting Units Raised to a Power
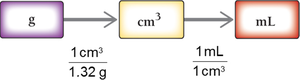
When converting units raised to a power, the conversion factor must also be raised to that power (e.g., cm3 to m3).
Physical Property: Density
Density is a fundamental property of matter, defined as mass per unit volume. It is used to compare substances and as a conversion factor between mass and volume.
Formula:
Units: g/cm3 or g/mL
Example: A liquid with a volume of 22.5 mL and a mass of 27.2 g has a density of
Density as a Conversion Factor
Density can be used to convert between mass and volume.
Review
Uncertainty: Last digit in a measurement is estimated.
Units: SI units are used; prefix multipliers help express measurements conveniently.
Density: Mass divided by volume; varies between substances.
Learning Outcomes
Express numbers in scientific notation.
Report measured quantities with correct digits.
Identify significant figures.
Round numbers appropriately.
Apply significant figure rules in calculations.
Convert between units, including those in numerator and denominator, and units raised to a power.
Calculate and use density as a conversion factor.
Highlight Problem Involving Units
Unit errors can have significant consequences. For example, NASA lost a $125 million Mars Climate Orbiter due to a unit conversion mistake between meters and feet.

Example: If one group specified 155 km as 1.55 × 105 m and another as 1.55 × 105 ft, the difference in altitude could cause mission failure.
