 Back
BackMolecular Orbital Theory and LCAO Models for Diatomic Molecules
Study Guide - Smart Notes

Atoms and Elements
Atomic Orbitals and Their Energies
Atomic orbitals (AOs) are regions in an atom where electrons are likely to be found. The energy of these orbitals depends on the atom and the type of orbital (s, p, etc.). - Key Point 1: The energy of atomic orbitals increases with distance from the nucleus and varies across the periodic table due to effective nuclear charge. - Key Point 2: The shape of orbitals (spherical for s, dumbbell for p) affects their overlap and bonding potential. - Example: The 1s orbital of hydrogen is higher in energy than the 2s orbital of lithium, which is much higher than the 1s orbital of lithium. 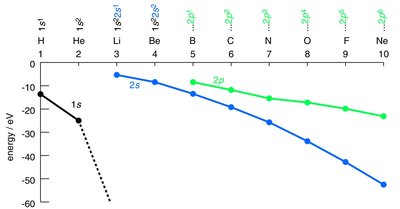
Molecules and Compounds
Linear Combination of Atomic Orbitals (LCAO) and Molecular Orbitals (MOs)
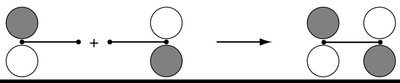
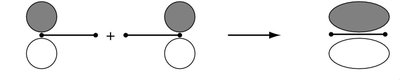
The LCAO method is used to construct molecular orbitals (MOs) by combining atomic orbitals from two atoms. This approach is fundamental to understanding chemical bonding in diatomic molecules. - Key Point 1: The number of MOs formed equals the number of AOs combined. - Key Point 2: Bonding MOs result from constructive (in-phase) overlap, while antibonding MOs result from destructive (out-of-phase) overlap. - Example: In H2, two 1s orbitals combine to form one bonding (σg) and one antibonding (σu*) MO. 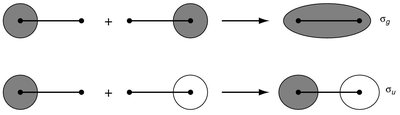
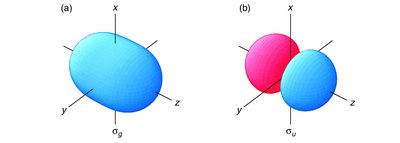
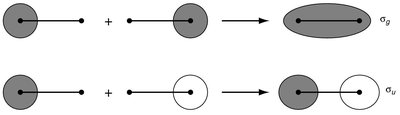
Bonding and Antibonding Molecular Orbitals
Bonding MOs are lower in energy than the original AOs, leading to stabilization. Antibonding MOs are higher in energy, causing destabilization. - Key Point 1: The extent of stabilization or destabilization depends on the degree of AO overlap. - Key Point 2: Only AOs of compatible symmetry and similar energy interact effectively. - Example: The σg MO has no nodal plane between atoms, while the σu* MO has a nodal plane. 
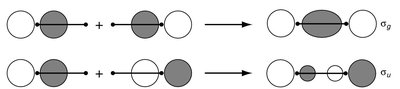
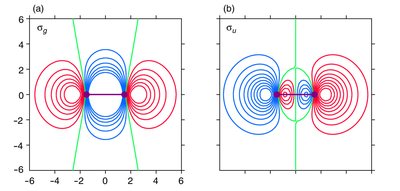
Chemical Bonding
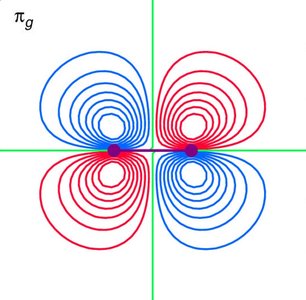
Symmetry and Overlap of p Orbitals
For diatomic molecules, p orbitals can overlap in different ways, leading to σ and π MOs. - Key Point 1: End-on overlap of p orbitals forms σ MOs; side-on overlap forms π MOs. - Key Point 2: Only orbitals of the correct symmetry interact to form MOs. - Example: In O2, the side-on overlap of 2p orbitals forms π MOs, which are responsible for the molecule's paramagnetism. 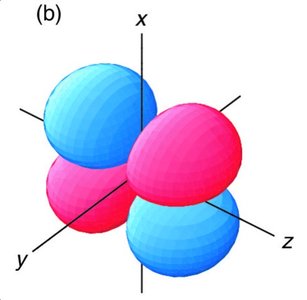
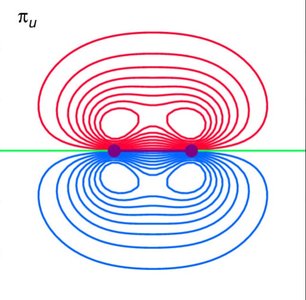
Molecular Orbital Energy Level Diagrams (MOELD)
MOELDs show the relative energies and occupancy of MOs in a molecule. - Key Point 1: Electrons fill MOs according to the Aufbau principle, Hund’s rule, and the Pauli exclusion principle. - Key Point 2: Bond order is calculated as . - Example: O2 has two unpaired electrons in π* orbitals, explaining its paramagnetism. 
Bond Order and Magnetic Properties
Bond order indicates the number of shared electron pairs and correlates with bond strength. Magnetic properties depend on the presence of unpaired electrons. - Key Point 1: Molecules with unpaired electrons are paramagnetic; those with only paired electrons are diamagnetic. - Key Point 2: Bond order can be used to predict molecular stability and reactivity. - Example: O2 is paramagnetic due to unpaired electrons in its MOELD. 
Polarization and Electronegativity in MO Formation
When combining AOs of different atoms, differences in energy and electronegativity lead to polarized MOs and increased ionic character. - Key Point 1: AOs closer in energy interact more strongly, leading to greater MO stabilization. - Key Point 2: The more electronegative atom contributes more to the MO, resulting in polarization. - Example: In LiH, the bonding MO is localized more on hydrogen due to its higher electronegativity. 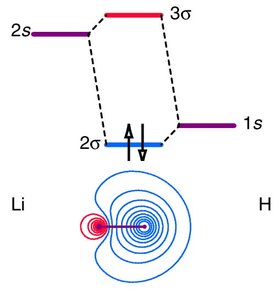
Overlap Between s and p Orbitals
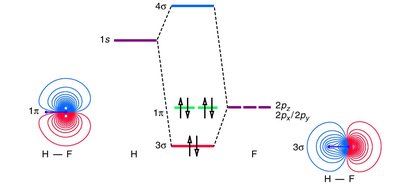
Overlap between s and p orbitals is symmetry-dependent. Only certain combinations are allowed. - Key Point 1: 1s/2pz overlap is symmetry forbidden; 1s/2px or 1s/2py overlap is allowed. - Key Point 2: The core 1s orbital of fluorine is too low in energy to interact with hydrogen’s 1s orbital in HF. - Example: The MOELD for HF shows non-bonding MOs due to energy mismatch. 
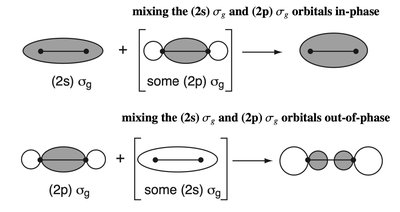
sp Mixing in Second Period Homodiatomics
In second period diatomic molecules, mixing between 2s and 2p orbitals (sp mixing) perturbs the MOELD. - Key Point 1: The degree of sp mixing increases with decreasing energy gap between 2s and 2p orbitals. - Key Point 2: This mixing affects the ordering and energies of MOs, especially in molecules like N2 and O2. - Example: The MOELD for N2 shows significant sp mixing, affecting bond order and molecular properties. 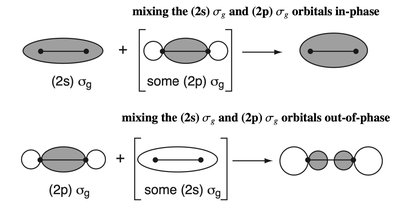
Advanced Bonding: Multiple Bonds and δ Bonds
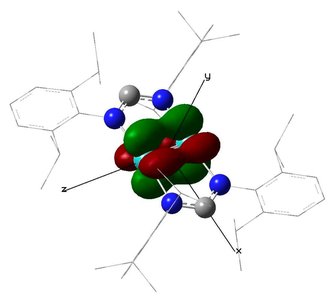
Some molecules exhibit bond orders greater than two, including rare quintuple bonds with σ, π, and δ components. - Key Point 1: δ bonds arise from side-on overlap of d orbitals, adding to the bond order. - Key Point 2: Molecules with high bond orders are experimentally known and have unique properties. - Example: The aurora borealis is related to ionized N2 emitting light upon returning to ground state. 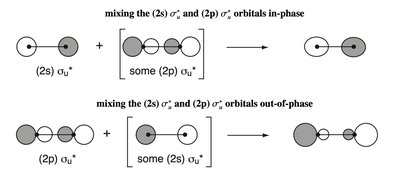
Summary
Key Takeaways
- The LCAO approach allows prediction of MO shapes and energies in diatomic molecules. - The limitations of the Lewis model are evident in molecules like O2, where MO theory explains paramagnetism. - Differences in electronegativity and AO energy lead to bond polarization and a continuum between covalent and ionic character. - MOELDs are essential for understanding molecular stability, magnetic properties, and advanced bonding scenarios.
Independent Exercise
Construct the MOELD for F2 using the given AO energies (2s, -40.0 eV; 2p, -18.1 eV). Confirm the bond order is 1 and sketch all MOs. Compare your answer with ChemTube3D resources. Additional info: This guide expands on lecture notes with academic context, definitions, and examples for clarity and completeness.
