 Back
BackMolecules and Compounds: Chemical Formulas, Nomenclature, and Types of Compounds
Study Guide - Smart Notes

Molecules and Compounds
Chemical Formulas
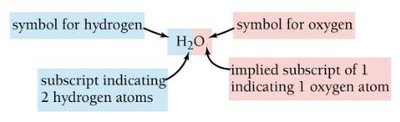
Chemical formulas are symbolic representations that show the elements and the number of atoms of each element in a compound. They provide essential information about the composition of both covalent and ionic compounds.
Covalent compounds: The formula shows the actual number of atoms of each element in a molecule.
Ionic compounds: The formula shows the ratio of atoms (or ions) in a formula unit.
Formulas are written with the most metallic element first.
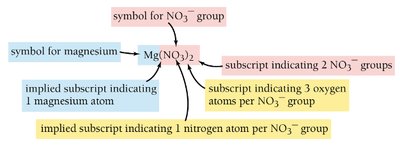
Parentheses are used in ionic formulas when more than one of a polyatomic ion is present.
Molecular vs. Empirical Formulas
The molecular formula shows the actual number of atoms of each element in a molecule, while the empirical formula shows the simplest whole-number ratio of atoms.
Molecular formula: Actual number of atoms (e.g., glucose: C6H12O6).
Empirical formula: Simplest ratio (e.g., glucose: CH2O).
For ionic compounds, the formula is always the smallest whole-number ratio of ions.
Formula Mass
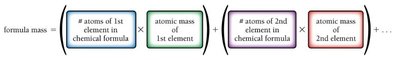
The formula mass is the average mass of the molecules or formula units that compose a compound. It is calculated as the sum of the atomic masses of all atoms in the chemical formula and is measured in atomic mass units (amu).
Example: CO2: (12.01 amu + 2 × 16.00 amu) = 44.01 amu/molecule
Example: CaCO3: (40.08 amu + 12.01 amu + 3 × 16.00 amu) = 100.10 amu/formula unit
The Law of Constant Composition (Law of Definite Proportions)
This law states that all samples of the same compound always contain the same proportions by mass of the component elements, regardless of sample size.
Example: Water always contains 8 g O for every 1 g H.
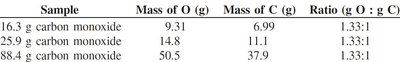
Example: CO always contains 2.66 g O for every 1 g C; CO2 always contains 1.33 g O for every 1 g C.


Sample | Mass of O (g) | Mass of C (g) | Ratio (g O : g C) |
|---|---|---|---|
123 g carbon dioxide | 89.4 | 33.6 | 2.66:1 |
50.5 g carbon dioxide | 36.7 | 13.8 | 2.66:1 |
88.6 g carbon dioxide | 64.4 | 24.2 | 2.66:1 |

Sample | Mass of O (g) | Mass of C (g) | Ratio (g O : g C) |
|---|---|---|---|
16.3 g carbon monoxide | 9.31 | 6.99 | 1.33:1 |
25.9 g carbon monoxide | 14.8 | 11.1 | 1.33:1 |
88.4 g carbon monoxide | 50.5 | 37.9 | 1.33:1 |
Types of Elements and Compounds
Elements: Atomic and Molecular
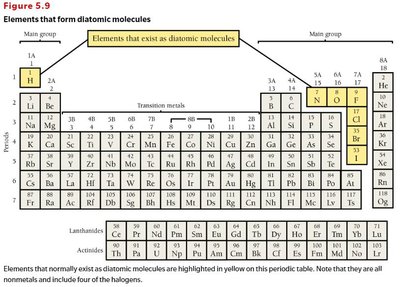
Elements can exist as single atoms (atomic) or as molecules (molecular).
Atomic elements: Exist as single atoms (e.g., Na, K, Mg, noble gases).
Molecular elements: Exist as molecules, often diatomic (e.g., H2, N2, O2, F2, Cl2, Br2, I2) or polyatomic (e.g., P4, S8).
Ionic Compounds (Salts)
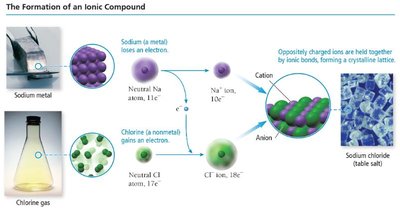
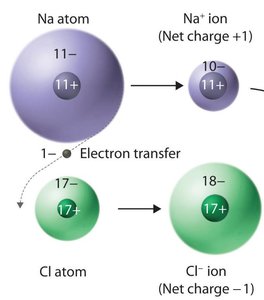
Ionic compounds are composed of cations and anions bound together by ionic bonds, which are electrostatic attractions between ions. They are generally formed by a combination of a metal and one or more nonmetals.
Cations: Formed by metals losing electrons.
Anions: Formed by nonmetals gaining electrons.
Formula units: The smallest electrically neutral collection of ions.

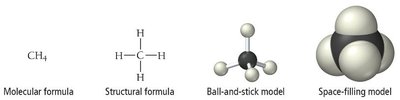
Molecular Compounds (Covalent Compounds)
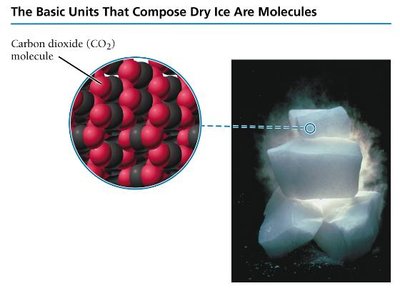
Molecular compounds consist of two or more covalently bonded atoms. Covalent bonds involve the sharing of electrons between atoms and are generally formed by two or more nonmetals.
Individual units: Called molecules.
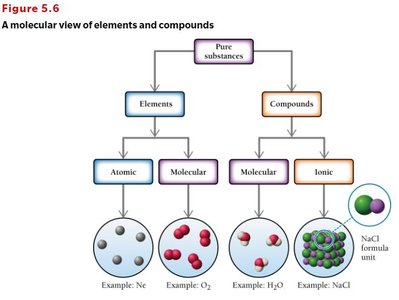
Monatomic and Polyatomic Ions
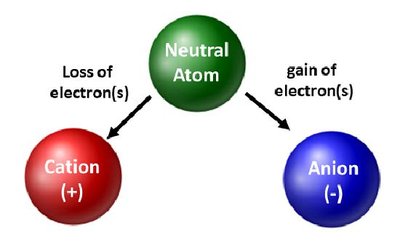
Monatomic Ions
Monatomic ions are single atoms with a positive or negative charge due to the loss or gain of electrons.
Cations: Positive charge, formed by loss of electrons.
Anions: Negative charge, formed by gain of electrons.
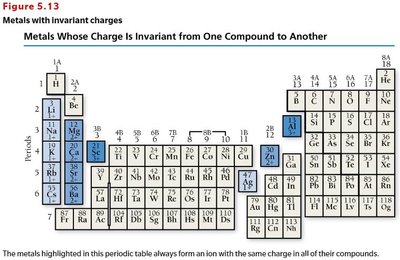
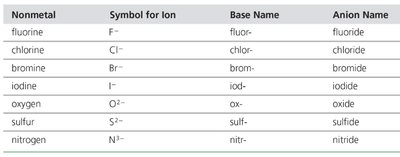
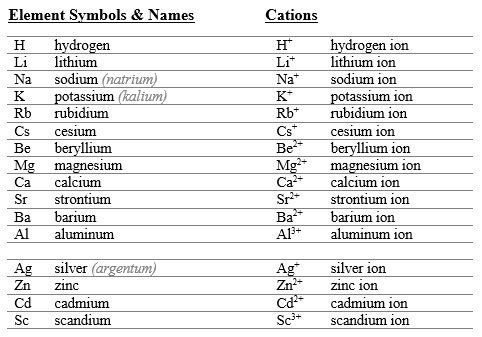
Systematic Nomenclature for Monatomic Ions
Monatomic ions are named systematically based on their charge and element type.
Cations (invariant charge): "element name + ion" (e.g., Na+ is sodium ion).
Cations (variant charge): "element name + (charge in Roman numerals) + ion" (e.g., Fe2+ is iron(II) ion).
Anions: "element root name + ide + ion" (e.g., O2- is oxide ion).
Polyatomic Ions
Polyatomic ions consist of two or more covalently bonded atoms with an overall charge.
Some polyatomic ions are named systematically, especially oxoanions (oxyanions), which contain a central atom and covalently bound oxygen atoms.
Oxoanion naming uses prefixes and suffixes to indicate the number of oxygen atoms:
# of oxygen atoms | Naming system |
|---|---|
Least | hypo- -ite ion |
Less | -ite ion |
More | -ate ion |
Most | per- -ate ion |
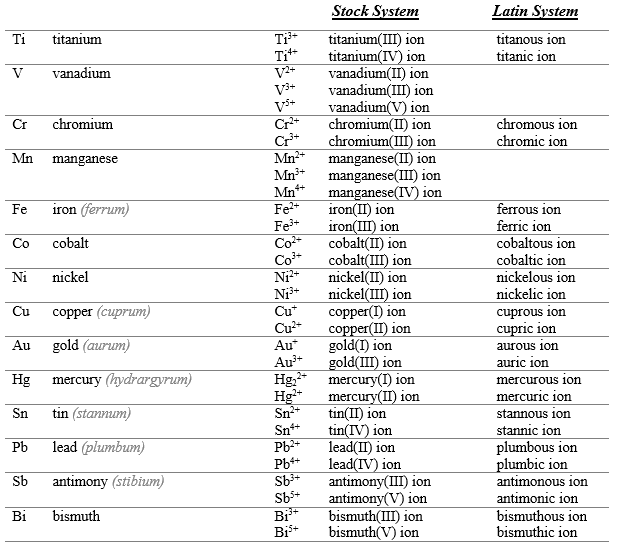
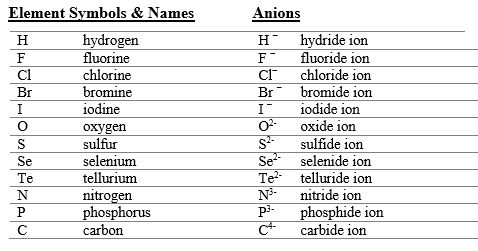
Systematic Nomenclature for Compounds
Writing Ionic Compound Formulas
To write ionic compound formulas:
Split the compound name into cation and anion names.
Convert names to formulas, including charges.
Combine ions in a ratio that forms a neutral unit.
Reduce ratios to the lowest terms.
Add subscripts to indicate the number of each ion; use parentheses for polyatomic ions.
Do not include ion charges in the final formula.
Naming Ionic Compounds
To name ionic compounds:
Break the formula into cation and anion formulas.
Name the cation and anion (removing "ion" from each).
Cation name is written first.
If the cation is a metal with multiple possible charges, indicate the charge in the name.
Hydrates
Hydrates and Their Nomenclature
Hydrates are ionic compounds with loosely bound water molecules. The water can be removed by heating, forming the anhydrate.
Name the ionic compound.
Add "hydrate" with a prefix to indicate the number of water molecules (e.g., monohydrate, hexahydrate).

Acids
Acid Nomenclature
Acids are covalent compounds that release hydrogen ions (H+) in water. Their names are based on the contained anion:
-ide ion: hydro- -ic acid (e.g., hydrochloric acid)
-ite ion: -ous acid (e.g., sulfurous acid)
-ate ion: -ic acid (e.g., sulfuric acid)
Binary Covalent Compounds
Nomenclature of Binary Covalent Compounds
Binary covalent compounds consist of two nonmetals. The first element is named first, the second element is named with the suffix -ide, and prefixes indicate the number of atoms.
Mono- is never used for the first element.
Prefixes: di-, tri-, tetra-, penta-, hexa-, etc.
Study Guide Summary
Know key terms: formula mass, molecular formula, empirical formula, atomic element, molecular element, compound, molecule, formula unit, ionic compound, ionic bond, monatomic ion, polyatomic ion, cation, anion, covalent compound, covalent bond, oxyanions, binary acid, polyatomic acid.
Be able to calculate formula mass using the periodic table.
Understand the law of constant composition.
Convert molecular formulas to empirical formulas.
Know element names and symbols, especially diatomic elements.
Distinguish between ionic and covalent compounds.
Apply systematic nomenclature for monatomic ions and oxyanions.
Convert between names and formulas for ionic compounds, hydrates, acids, and binary covalent compounds.
Additional info: This guide covers all major topics from Chapter 5, including chemical formulas, types of compounds, nomenclature, and the law of constant composition, as well as practical examples and tables for reference.
