 Back
BackMolecules and Compounds: Structure, Classification, and Nomenclature
Study Guide - Smart Notes

Molecules and Compounds
The Mole and Avogadro's Number
The concept of the mole is fundamental in chemistry for quantifying the amount of substance. A mole represents a specific number of particles, known as Avogadro's Number, which is particles. This allows chemists to work with manageable quantities when dealing with atoms and molecules, which are extremely small and numerous.
Avogadro's Number: The number of particles in one mole, defined as the number of atoms in exactly 12 grams of carbon-12.
Application: Used to convert between the number of particles and the amount of substance in moles.

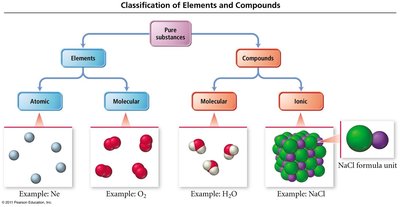
Classification of Elements and Compounds
Chemical substances are classified as either elements or compounds. Elements can exist as single atoms (atomic) or as molecules (molecular). Compounds are further divided into molecular compounds (composed of molecules) and ionic compounds (composed of ions).
Elements: Pure substances consisting of only one type of atom.
Atomic Elements: Exist as single atoms (e.g., Ne).
Molecular Elements: Exist as molecules (e.g., O2).
Compounds: Pure substances composed of two or more elements.
Molecular Compounds: Made of molecules (e.g., H2O).
Ionic Compounds: Made of ions (e.g., NaCl).
Molecules of the Elements
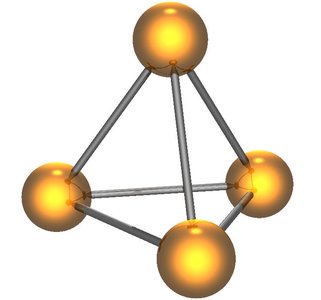
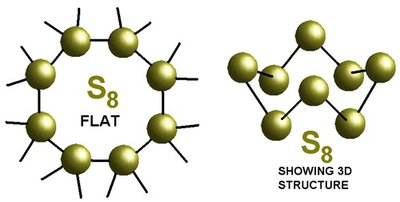
Diatomic, Tetratomic, and Octatomic Molecules
Some elements exist naturally as molecules rather than individual atoms. The most common are diatomic molecules (two atoms), but some elements form tetratomic (four atoms) or octatomic (eight atoms) molecules.
Diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2
Tetratomic molecules: P4 (phosphorus)
Octatomic molecules: S8 (sulfur), Se8 (selenium)


Formation and Classification of Compounds
Predicting Cations and Anions
Ionic compounds are formed between metals and nonmetals. Metals tend to lose electrons and become cations (positively charged), while nonmetals gain electrons to become anions (negatively charged). Main group elements gain or lose electrons to achieve the same number of electrons as their nearest noble gas.
Cations: Formed by metals losing electrons.
Anions: Formed by nonmetals gaining electrons.
Periodic Table Trends: Group 1A (+1), 2A (+2), 3A (+3), 5A (−3), 6A (−2), 7A (−1).

Covalent Compounds
Covalent bonds are formed by sharing electrons between two atoms, usually nonmetals. Each atom contributes one electron, and the shared electrons are mutually attracted to both nuclei, shielding them from repulsive forces.
Bond Formation: Occurs when sharing electrons lowers energy.
Bond Length: Optimum distance between nuclei maximizes stabilization.
Repulsion: Electrons and protons repel, but net attractive force dominates.

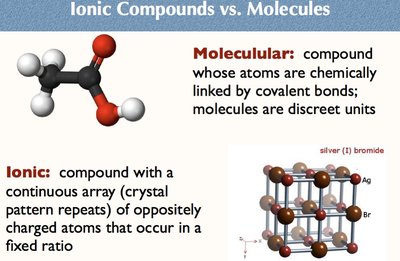
Ionic Compounds vs. Molecular Compounds
Molecular compounds consist of discrete molecules linked by covalent bonds. Ionic compounds have a continuous array of oppositely charged ions in a fixed ratio, forming a crystal lattice.
Molecular: Discrete units, covalent bonds.
Ionic: Crystal lattice, fixed ratio, electrostatic attraction.
Chemical Formulas
Types of Chemical Formulas
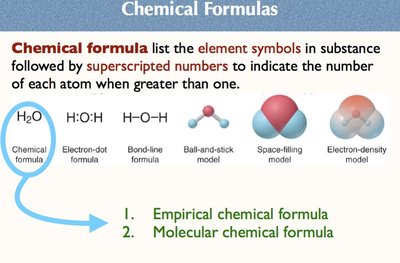
A chemical formula lists the element symbols in a substance, followed by subscripts indicating the number of each atom. There are several ways to represent molecules, including empirical and molecular formulas.
Empirical formula: Shows the simplest whole-number ratio of atoms.
Molecular formula: Shows the actual number of atoms in a molecule.
Other representations: Electron-dot, bond-line, ball-and-stick, space-filling, electron-density models.
Subscripts and Coefficients
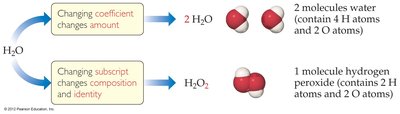
Subscripts and coefficients in chemical formulas provide different information. Subscripts indicate the number of atoms of each element in a molecule, while coefficients indicate the number of molecules.
Subscript: Changes composition and identity (e.g., H2O vs. H2O2).
Coefficient: Changes the amount (e.g., 2 H2O means two molecules).
Naming Compounds
Naming Ionic Compounds
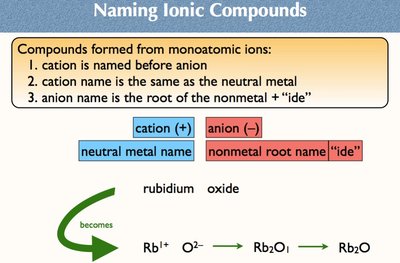
Ionic compounds formed from monoatomic ions follow specific naming rules:
Cation is named before anion.
Cation name: Same as the neutral metal.
Anion name: Root of the nonmetal + "ide".
Polyatomic Ions
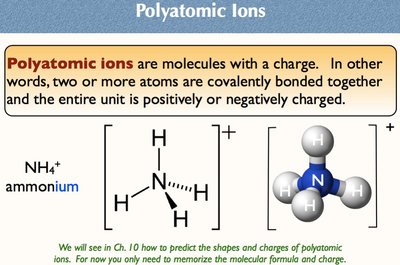
Polyatomic ions are molecules with a charge, consisting of two or more covalently bonded atoms. The entire unit is positively or negatively charged.
Example: Ammonium ion (NH4+).
Importance: Memorize molecular formula and charge.
Naming Binary Covalent Compounds
Binary covalent compounds are formed from two nonmetals. The naming convention uses prefixes to indicate the number of atoms and ends the second element with "-ide".
Prefix + name of 1st element
Prefix + base name of 2nd element + "-ide"
Examples: H2O (water), CH4 (methane), NH3 (ammonia), PH3 (phosphine)


Naming Oxanions
Oxanions are negatively charged polyatomic ions containing oxygen. Families are named systematically according to the number of oxygen atoms.
Most O atoms: Suffix "-ate" (e.g., sulfate SO42−).
Least O atoms: Suffix "-ite" (e.g., sulfite SO32−).

Naming Hydrates
Hydrates are ionic compounds that contain water molecules within their crystal structure. The name includes a prefix indicating the number of water molecules and the term "hydrate".
Example: CoCl2·6H2O is cobalt(II) chloride hexahydrate.
Anhydrous: Compound without water (e.g., CoCl2).

Summary Table: Types of Compounds and Naming Conventions
Type | Example | Naming Rule |
|---|---|---|
Atomic Element | Ne | Element name |
Molecular Element | O2 | Element name |
Molecular Compound | H2O | Prefix + element + prefix + element + "-ide" |
Ionic Compound | NaCl | Cation name + anion root + "ide" |
Polyatomic Ion | NH4+ | Memorize formula and charge |
Oxanion | SO42− | "-ate" or "-ite" suffix |
Hydrate | CoCl2·6H2O | Compound name + prefix + "hydrate" |
Additional info: This guide expands on brief lecture points to provide full academic context, definitions, and examples for each topic, ensuring completeness and clarity for exam preparation.
