 Back
BackMolecules and Compounds: Structure, Types, and Nomenclature
Study Guide - Smart Notes

Molecules & Compounds
Organizing Matter
Understanding matter at the particle level is essential in chemistry. Matter can be organized based on the types of particles present and how they are combined. The vocabulary of chemistry—such as atoms, molecules, and compounds—helps describe these combinations.
Atoms: The basic units of chemical elements.
Molecules: Groups of two or more atoms bonded together.
Compounds: Substances composed of at least two different types of atoms in a fixed ratio.
Compounds can be represented by chemical formulas, which show the types and ratios of atoms present.
Compounds and Chemical Formulas
Compounds always have a consistent ratio of their constituent atoms, which is reflected in their chemical formulas. These formulas use subscripts to indicate the number of each type of atom and may use parentheses and coefficients to show multiples.
Subscripts indicate the number of atoms of each element in a molecule (e.g., H2O).
Coefficients multiply the entire formula (e.g., 4 H2O2 means 4 molecules of hydrogen peroxide).
Parentheses are used when a group of atoms appears more than once (e.g., (NH4)2O).
Practice interpreting formulas by counting the number of each atom present in a given formula.
Types of Compounds
Ionic vs. Molecular Compounds
Compounds can be classified as ionic or molecular based on the types of elements involved and the nature of their bonding.
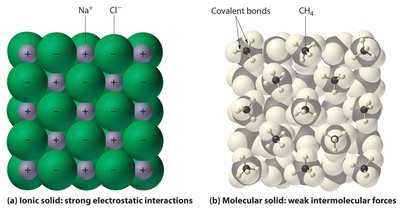
Ionic Compounds: Formed from metals and nonmetals; consist of positive (cation) and negative (anion) ions held together by strong electrostatic forces in a crystal lattice. The smallest ratio of ions is called a "formula unit."
Molecular Compounds: Formed from nonmetals; consist of discrete molecules held together by covalent bonds. Intermolecular forces between molecules are generally weaker than ionic bonds.
Binary Compounds
Binary compounds contain only two different elements. They can be either molecular or ionic.
Molecular Binary Compounds: Usually consist of two nonmetals.
Ionic Binary Compounds: Consist of a metal and a nonmetal.
Determining Compound Type by Formula
To determine if a compound is ionic or molecular, examine the first element in the formula:
If it starts with a nonmetal, it is probably molecular (exceptions: ammonium ion, some metalloids).
If it starts with a metal, it is ionic.
Naming Compounds
Naming Molecular Binary Compounds
Molecular binary compounds use prefixes to indicate the number of each atom. The general pattern is:
prefix + name of first element + prefix + root of second element + "-ide"
"Mono-" is never used for the first element.
Examples:
PCl3: phosphorus trichloride
N2O7: dinitrogen heptoxide
BrF: bromine monofluoride
Naming Ionic Binary Compounds
Ionic binary compounds are named by stating the cation (metal) first, followed by the anion (nonmetal) with an "-ide" ending.
Cation name + Anion name
For transition metals with multiple possible charges, use Roman numerals to indicate the charge (IUPAC standard).
Examples:
AlCl3: aluminum chloride
FeCl3: iron (III) chloride
CaI2: calcium iodide
Polyatomic Ions
Polyatomic ions are groups of covalently bonded atoms that carry a charge. Many common polyatomic ions contain oxygen and are called oxyanions.
The most common form gets the "-ate" ending; one fewer oxygen gets "-ite." Prefixes like "per-" (more oxygen) and "hypo-" (less oxygen) are also used.
Ion | Formula | Name |
|---|---|---|
SO42- | Sulfate | |
SO32- | Sulfite | |
ClO3- | Chlorate | |
ClO2- | Chlorite |

Polyatomic Cations
NH4+: ammonium
Hg22+: mercury (I) ion (less common)
Writing Formulas
Writing Molecular Formulas
Prefixes in the name tell you the number of each atom. Write the formula accordingly.
Dihydrogen monoxide: H2O
Tricarbon octahydride: C3H8
Phosphorus trichloride: PCl3
Writing Ionic Formulas
Ionic formulas must balance the total positive and negative charges. Use the criss-cross method or algebraic balancing.
Calcium chloride: CaCl2
Sodium sulfate: Na2SO4
Ammonium phosphate: (NH4)3PO4
Naming Acids
Oxoacids
Oxoacids are acids that contain hydrogen, oxygen, and another element (the central atom). Their names are based on the oxyanion present:
If the anion ends in "-ate," the acid name ends in "-ic acid" (e.g., H2SO4: sulfuric acid).
If the anion ends in "-ite," the acid name ends in "-ous acid" (e.g., H2SO3: sulfurous acid).
Prefixes "per-" and "hypo-" are retained (e.g., HClO4: perchloric acid; HClO: hypochlorous acid).
Binary Acids
Binary acids consist of hydrogen and one other nonmetal. Their names follow the pattern: "hydro-" + root of nonmetal + "-ic acid" (e.g., HCl: hydrochloric acid).
Summary Table: Naming Patterns
Type | Formula Example | Name Example |
|---|---|---|
Molecular Binary | N2O5 | dinitrogen pentoxide |
Ionic Binary | NaCl | sodium chloride |
Oxoacid | HNO3 | nitric acid |
Binary Acid | HBr | hydrobromic acid |
Practice Problems
Sulfuric acid: H2SO4
HFO: hypofluorous acid
H3PO3: phosphorous acid
HBr: hydrobromic acid
Perbromic acid: HBrO4
HClO3: chloric acid
