Back
BackNaming Chemical Compounds: Ionic, Molecular, and Acids
Study Guide - Smart Notes

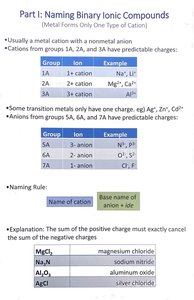
Naming Binary Ionic Compounds
Overview
Binary ionic compounds are formed from a metal cation and a nonmetal anion. The naming conventions depend on the predictability of the cation's charge and the identity of the anion.
Metal cations from groups 1A, 2A, and 3A have predictable charges.
Anions from groups 5A, 6A, and 7A also have predictable charges.
Some transition metals only have one charge (e.g., Ag+, Zn2+, Cd2+).
Predictable Charges
Group | Ion | Example |
|---|---|---|
1A | +1 cation | Na+ |
2A | +2 cation | Mg2+ |
3A | +3 cation | Al3+ |
Group | Ion | Example |
|---|---|---|
5A | -3 anion | N3- |
6A | -2 anion | O2- |
7A | -1 anion | Cl- |
Naming Rule
Name of cation + base name of anion + -ide
Charge Balance
The sum of the positive charges must exactly cancel the sum of the negative charges in the compound.
Formula | Name |
|---|---|
MgCl2 | magnesium chloride |
Na3N | sodium nitride |
Al2O3 | aluminum oxide |
AgCl | silver chloride |
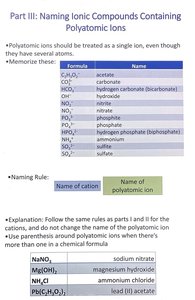
Naming Ionic Compounds Containing Polyatomic Ions
Overview
Polyatomic ions are groups of atoms that act as a single ion. They must be memorized and treated as a unit when naming compounds.
Common Polyatomic Ions
Formula | Name |
|---|---|
C2H3O2- | acetate |
CO32- | carbonate |
HCO3- | hydrogen carbonate (bicarbonate) |
OH- | hydroxide |
NO3- | nitrate |
NO2- | nitrite |
PO43- | phosphate |
PO33- | phosphite |
NH4+ | ammonium |
SO42- | sulfate |
SO32- | sulfite |
Naming Rule
Name of cation + name of polyatomic ion
Examples
Formula | Name |
|---|---|
NaNO3 | sodium nitrate |
Mg(OH)2 | magnesium hydroxide |
NH4Cl | ammonium chloride |
Pb(C2H3O2)2 | lead(II) acetate |
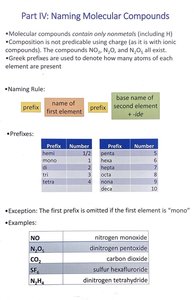
Naming Molecular Compounds
Overview
Molecular compounds are composed only of nonmetals. Their composition is not predictable using charge, so Greek prefixes are used to indicate the number of atoms of each element present.
Naming Rule
Prefix + name of first element + prefix + base name of second element + -ide
Common Prefixes
Prefix | Number |
|---|---|
mono | 1 |
di | 2 |
tri | 3 |
tetra | 4 |
penta | 5 |
hexa | 6 |
hepta | 7 |
octa | 8 |
nona | 9 |
deca | 10 |
Exceptions
The first prefix is omitted if the first element is "mono".
Examples
Formula | Name |
|---|---|
NO | nitrogen monoxide |
NO2 | dinitrogen monoxide |
CO2 | carbon dioxide |
SF4 | sulfur tetrafluoride |
N2H4 | dinitrogen tetrahydride |
Naming Binary Acids
Overview
Binary acids consist of hydrogen and a nonmetal (not polyatomic). The acid is named based on the nonmetal.
Naming Rule
hydro + base name of nonmetal + -ic + acid
Examples
Formula | Name |
|---|---|
HCl (aq) | hydrochloric acid |
HF (aq) | hydrofluoric acid |
H2S (aq) | hydrosulfuric acid |
Exception
This rule applies only when the acid is dissolved in water (aqueous, aq). If not dissolved, name it using ionic compound rules.
Formula | Name |
|---|---|
HCl (aq) | hydrochloric acid (dissolved in water) |
HCl (g) | hydrogen chloride (pure substance) |

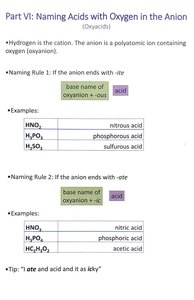
Naming Acids with Oxygen in the Anion (Oxyacids)
Overview
Oxyacids contain hydrogen and a polyatomic ion with oxygen (oxyanion). The naming depends on the ending of the oxyanion.
Naming Rule 1: If the anion ends with -ite
base name of oxyanion + -ous + acid
Examples
Formula | Name |
|---|---|
HNO2 | nitrous acid |
H3PO3 | phosphorous acid |
H2SO3 | sulfurous acid |
Naming Rule 2: If the anion ends with -ate
base name of oxyanion + -ic + acid
Examples
Formula | Name |
|---|---|
HNO3 | nitric acid |
H3PO4 | phosphoric acid |
HC2H3O2 | acetic acid |
Tip
"-ate" and "acid" rhyme as "-ic".