 Back
BackNuclear Radiation: Introductory Chemistry Study Guide
Study Guide - Smart Notes

Nuclear Radiation
Natural Radioactivity
Natural radioactivity refers to the spontaneous emission of radiation from certain unstable atomic nuclei. This process occurs in nature and is responsible for the presence of radioactive elements in the environment.
Radioactivity: The process by which unstable nuclei lose energy by emitting radiation.
Isotope: Atoms of the same element with different numbers of neutrons, resulting in different mass numbers.
Radioisotope: An isotope that emits radiation.
Example: Carbon-14 (146C) is a radioisotope used in radiocarbon dating.
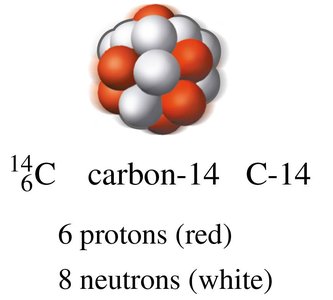
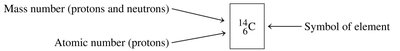
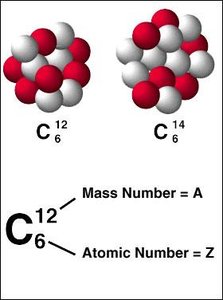
Stability of Nuclei
The stability of a nucleus depends on the ratio of protons to neutrons. Stable nuclei have balanced forces, while unstable nuclei have too many or too few protons or neutrons, leading to radioactivity.
Stable Isotopes: Most elements with atomic numbers less than 20 have stable isotopes.
Unstable Isotopes: Elements with atomic numbers greater than 20 often have one or more unstable isotopes.



Types of Radiation
Radioactive decay can emit several types of radiation, each with distinct properties and effects.
Alpha (α) particles: Consist of 2 protons and 2 neutrons; represented as 42He or α. They have a charge of +2 and are relatively heavy.
Beta (β) particles: High-energy electrons (0-1e) or positrons (0+1e); lighter and can penetrate further than alpha particles.
Gamma (γ) rays: Pure energy, no mass or charge; highly penetrating.

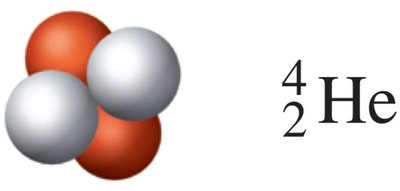



Type | Symbol | Charge | Penetration |
|---|---|---|---|
Alpha | or | +2 | Low |
Beta | or | -1 | Medium |
Positron | or | +1 | Medium |
Gamma | or | 0 | High |
Radioactive Decay and Nuclear Equations
Radioactive decay is the process by which an unstable nucleus emits radiation and transforms into a more stable nucleus. Nuclear equations represent these transformations.
Alpha Decay: Loss of an alpha particle. Example:
Beta Decay: Loss of a beta particle. Example:
Positron Emission: Loss of a positron. Example:
Gamma Emission: Release of gamma rays. Example:
Radiation Measurement
Radiation is measured in units that quantify the amount of radioactive decay and its effects on living organisms.
Curie (Ci): 3.7 x 1010 disintegrations per second.
Gray (Gy): Absorbed dose of radiation.
Sievert (Sv): Biological effect of radiation.
Rem: Roentgen equivalent man; 1 Gy = 1 Sv = 100 rem.
Half-Life of a Radioisotope
The half-life is the time required for half of the radioactive atoms in a sample to decay. This property is used to determine the age of materials and the duration of radioactivity in medical applications.
Formula:
Example: If 500 mg of Chromium-51 (t1/2 = 28 days) is present, after 56 days (2 half-lives), 125 mg remains.
Medical Applications Using Radioactivity
Radioisotopes are used in medicine for diagnosis and treatment. Common applications include cancer treatment, brain scans, blood flow studies, bone scans, and lung infection detection.
Medical Use | Atomic Symbol | Mass Number | Number of Protons | Number of Neutrons |
|---|---|---|---|---|
Cancer treatment | 131 | 55 | 76 | |
Brain scan | 99 | 43 | 56 | |
Blood flow | 141 | 58 | 83 | |
Bone scan | 85 | 47 | 38 | |
Lung infection | 133 | 54 | 79 |

Nuclear Fission and Fusion
Nuclear fission and fusion are processes that release large amounts of energy by splitting or combining atomic nuclei.
Fission: Splitting of a heavy nucleus into smaller nuclei, releasing energy. Example:
Fusion: Combining of two light nuclei to form a heavier nucleus, releasing energy. Example:
Shielding and Safety
Shielding is used to prevent exposure to harmful radiation. Materials such as lead, concrete, and water are commonly used to absorb or block radiation.
Additional info:
Radioactivity is a fundamental concept in nuclear chemistry and is essential for understanding nuclear reactions, medical imaging, and energy production.
Radiation safety is critical in both medical and industrial applications to protect human health.
