 Back
BackOxidation and Reduction (Redox) Chemistry: Study Notes
Study Guide - Smart Notes

Ch.16 Oxidation and Reduction
Calculate Oxidation Numbers
Oxidation numbers (or oxidation states) are used to keep track of electron transfer in chemical reactions, especially in redox processes. They help determine which elements are oxidized and which are reduced.
Oxidation Number: A value assigned to an element in a compound or ion that represents its ability to gain, lose, or share electrons.
Natural State: For an atom in its elemental (standard) state, the oxidation number is zero. For example, O2, H2, N2, and all pure elements have an oxidation number of 0.
Ions: For a monoatomic ion, the oxidation number is equal to its charge. For example, Na+ has an oxidation number of +1, Cl- is -1.
Example: In the compound NaCl, Na has an oxidation number of +1 and Cl is -1.
Practice: Noble gases like Argon (Ar) in their elemental state have an oxidation number of 0.
Example: Compounds like O2 or He have oxidation numbers of zero for each atom.
Oxidation Number Rules
Oxidation numbers are assigned using a set of rules, as they do not always correspond to actual charges in covalent compounds.
Assign known oxidation numbers to listed elements (e.g., Group 1A is +1, Group 2A is +2, etc.).
For non-listed elements, treat the unknown as a variable and solve using the sum of oxidation numbers equal to the compound's charge.
Distribute oxidation numbers according to subscripts in the formula.
Example: In the acetate ion (C2H3O2-), assign known values to H and O, then solve for C.
Redox Reactions (Oxidation-Reduction Reactions)
Redox reactions involve the transfer of electrons between substances. One species is oxidized (loses electrons), and another is reduced (gains electrons).
Oxidizing Agent: The substance that is reduced (gains electrons).
Reducing Agent: The substance that is oxidized (loses electrons).
Mnemonic: LEO the lion says GER (Lose Electrons = Oxidation, Gain Electrons = Reduction).

Oxidation: Becomes more positive in charge; oxidation number increases.
Reduction: Becomes more negative in charge; oxidation number decreases.
Example: In the reaction 2 Li (s) + Zn2+ (aq) → 2 Li+ (aq) + Zn (s), Li is oxidized and Zn2+ is reduced.
Spontaneous Redox Reactions and the Activity Series
Some redox reactions occur spontaneously, meaning one element can displace another from a compound. The activity series ranks elements by their tendency to lose electrons (be oxidized).
An element higher in the activity series will displace one lower in the series.
The strongest reducing agents are at the top; the strongest oxidizing agents are at the bottom.
Example: Ca (s) + AgCl (s) → CaCl2 + Ag (s) is spontaneous because Ca is above Ag in the activity series.
Balancing Redox Reactions: Acidic Solutions
Balancing redox reactions in acidic solutions involves accounting for both mass and charge, using H+ and H2O as needed.
Break the reaction into two half-reactions (oxidation and reduction).
Balance all elements except H and O.
Balance O by adding H2O.
Balance H by adding H+.
Balance charge by adding electrons to the more positive side.
Multiply half-reactions to equalize electrons, then add and cancel intermediates.
Example: Balance NO2- (aq) + Cr2O72- (aq) → Cr3+ (aq) + NO3- (aq) in acidic solution.
Balancing Redox Reactions: Basic Solutions
Balancing in basic solutions follows the same steps as acidic, with an additional step to neutralize H+ by adding OH- to both sides, forming H2O.
Break into half-reactions and balance as in acidic solution.
Add OH- to both sides for each H+ present.
Combine H+ and OH- to form H2O, and cancel intermediates as needed.
Example: Balance MnO4- (aq) + N2H4 (aq) → Mn2+ (aq) + NO3- (aq) in basic solution.
Galvanic (Voltaic) Cells
A galvanic cell is an electrochemical cell that generates electricity from a spontaneous redox reaction. It consists of two half-cells connected by a salt bridge and external circuit.
Anode: Site of oxidation (loss of electrons); electrons flow from anode to cathode.
Cathode: Site of reduction (gain of electrons).
Salt Bridge: Allows ions to flow and maintain electrical neutrality.
Voltmeter: Measures the cell's voltage.
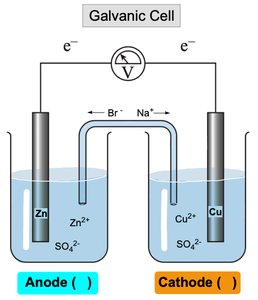
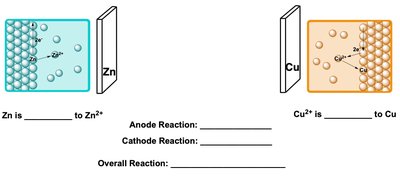
Example: In a Zn/Cu cell, Zn is oxidized at the anode and Cu2+ is reduced at the cathode.
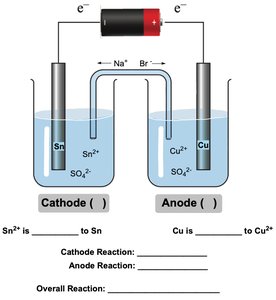
Electrolytic Cells
Electrolytic cells use external electrical energy to drive nonspontaneous redox reactions (electrolysis). The cathode is negative and the anode is positive, opposite to galvanic cells.
Electrolysis: The process of using electricity to force a chemical change.
Applications: Rechargeable batteries, electroplating, and lithium battery charging.
In all cells, reduction occurs at the cathode and oxidation at the anode.
Key Comparisons: Galvanic vs. Electrolytic Cells
Feature | Galvanic Cell | Electrolytic Cell |
|---|---|---|
Spontaneity | Spontaneous | Nonspontaneous (requires external energy) |
Energy Conversion | Chemical to electrical | Electrical to chemical |
Anode | Negative (site of oxidation) | Positive (site of oxidation) |
Cathode | Positive (site of reduction) | Negative (site of reduction) |
Summary Table: Redox Terminology
Term | Definition |
|---|---|
Oxidation | Loss of electrons; increase in oxidation number |
Reduction | Gain of electrons; decrease in oxidation number |
Oxidizing Agent | Substance that is reduced (gains electrons) |
Reducing Agent | Substance that is oxidized (loses electrons) |
Key Equations
General redox half-reaction (oxidation):
General redox half-reaction (reduction):
Additional info: These notes cover the essential concepts of oxidation numbers, redox reactions, balancing redox equations in acidic and basic solutions, and the operation of galvanic and electrolytic cells, as required for an introductory college chemistry course.
