 Back
BackPolyatomic Ions, Acids, Bases, and Solution Chemistry: Essential Concepts for Introductory Chemistry
Study Guide - Smart Notes

Polyatomic Ions
Definition and Structure
Polyatomic ions are fundamental chemical species encountered in introductory chemistry. They consist of two or more atoms covalently bonded, carrying a net positive or negative charge, and act as a single unit in chemical reactions.
Definition: A polyatomic ion is a charged group of covalently bonded atoms.
Net Charge: The entire group behaves as a single ion with a net charge, despite containing multiple atoms.
Nomenclature: Polyatomic ions often have names ending in -ate or -ite (e.g., sulfate, nitrate).
Example: Sulfate (SO42-), Nitrate (NO3-), Carbonate (CO32-)
Occurrence in Compounds
Polyatomic ions are commonly found in metal salts and acids.
Metal Salts: Examples include copper sulfate, magnesium carbonate, and iron nitrate.
Acids: Sulfuric acid, phosphoric acid, and nitric acid contain polyatomic ions.
Acids and Bases
Arrhenius Definition
The Arrhenius definition is a classical approach to acids and bases, focusing on their behavior in aqueous solutions.
Acid: Increases the concentration of hydrogen ions (H+) in water.
Base: Increases the concentration of hydroxide ions (OH-) in water.
Example: Hydrochloric acid (HCl) dissociates to produce H+ ions; sodium hydroxide (NaOH) produces OH- ions.
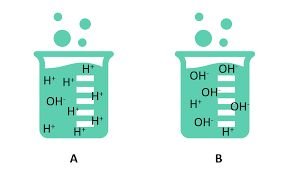
Acidic, Basic, and Neutral Solutions
The relative concentrations of H+ and OH- ions determine whether a solution is acidic, basic, or neutral.
Acidic: More H+ ions than OH- ions.
Basic: More OH- ions than H+ ions.
Neutral: Equal concentrations of H+ and OH- ions.
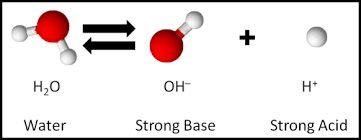
Dissociation of Water
Water undergoes a slight dissociation, producing equal amounts of H+ and OH- ions, making it neutral.
Equation:
Concentration: In pure water, [H+] = 0.0000001 mol/dm3 (1 x 10-7 mol/L).
The Mole Concept
Definition and Avogadro's Number
The mole is a fundamental unit in chemistry, representing a specific number of particles.
Definition: One mole contains 6.02 x 1023 particles (Avogadro's number).
Application: 1 mole of hydrogen atoms, oxygen atoms, water molecules, or copper(II) sulfate pentahydrate all contain the same number of particles.
pH and Acidity
Definition of pH
pH is a scale used to measure the acidity or alkalinity of a solution, based on the concentration of hydrogen ions.
Formula:
Interpretation: A pH of 7 is neutral; values below 7 are acidic, and above 7 are basic (alkaline).
Logarithmic Scale: The pH scale compresses large ranges of [H+] into manageable numbers.
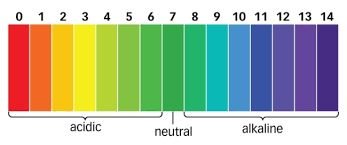
pH and Hydrogen Ion Concentration
pH 1: [H+] = 0.1 mol/dm3
pH 10: [H+] = 0.0000000001 mol/dm3
Acidity & Dissociation
Acidity as Hydrogen Ion Concentration
Acidity is determined by the concentration of free hydrogen ions in a solution.
Dissociation Process of Acids
When acids dissolve in water, they dissociate into ions.
Strong Acids: Ionize completely (100%), producing maximum H+ ions. The reaction is irreversible and results in high conductivity.
Weak Acids: Ionize partially (<5%), establishing equilibrium with most molecules intact. Conductivity is lower.
Example: Sulfuric acid (H2SO4) is a strong acid; acetic acid (CH3COOH) is a weak acid. 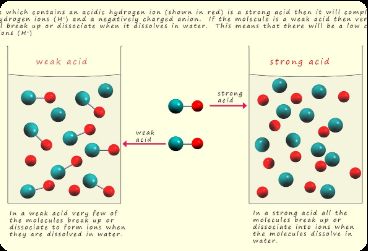
Concentration vs. Strength
Definitions and Distinctions
Strength: Refers to the degree of ionization of an acid or base (strong = complete, weak = partial).
Concentration: Refers to the amount of solute per unit volume (molarity, M).
Key Point: A strong acid can be dilute, and a weak acid can be concentrated.
Changing Concentration: Dilution
Dilution Process
Dilution is the process of reducing the concentration of a solution by adding solvent (usually water).
Stock Solution: The original, more concentrated solution.
Final Solution: The diluted solution after adding solvent.
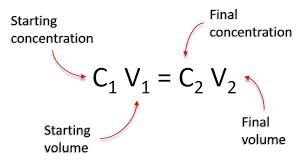
Formula: Where C1 = initial concentration, V1 = initial volume, C2 = final concentration, V2 = final volume.

Summary Table: Strong vs. Weak Acids
Property | Strong Acid | Weak Acid |
|---|---|---|
Degree of Ionization | 100% (Complete) | <5% (Partial) |
Conductivity | High | Low |
Reaction | Irreversible | Equilibrium |
Example | Sulfuric Acid (H2SO4) | Acetic Acid (CH3COOH) |
Summary Table: Concentration vs. Strength
Property | Concentration | Strength |
|---|---|---|
Definition | Amount of solute per unit volume | Degree of ionization |
Units | Molarity (mol/L) | Percentage (%) |
Example | 0.1 M HCl | Strong acid (HCl), Weak acid (CH3COOH) |
Practice and Application
Worksheet and Answer Key
Practice problems and answer keys are essential for mastering these concepts. Additional info: The worksheet and answer key are referenced but not included in the notes.
