Back
BackPolymers: Structure, Types, and Applications
Study Guide - Smart Notes

Polymers
Introduction to Polymers
Polymers are large macromolecules composed of repeating structural units called monomers. The term "polymer" is derived from the Greek words poly (many) and meros (part). Polymers can have molecular weights in the thousands and are essential in both natural and synthetic materials.
Monomer: The small, repeating unit that joins to form a polymer.
Macromolecule: A very large molecule, such as a polymer, with a high molecular weight.
Natural Polymers
Many substances found in nature are polymers. These include:
Starch
Cotton (cellulose)
Wood (cellulose)
Proteins

Early Synthetic Polymers
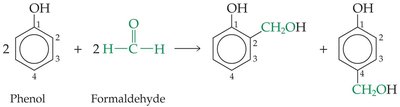
Celluloid, produced by treating natural cellulose with nitric acid, was one of the earliest synthetic polymers. It was used for billiard balls, men’s collars, and movie film. The first completely synthetic polymers were phenol–formaldehyde resins.
Types of Synthetic Polymers
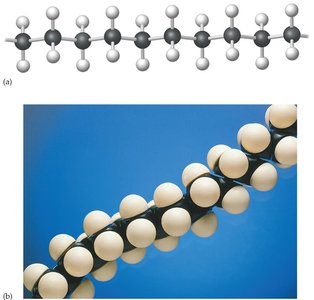
Polyethylene
Polyethylene is one of the most common synthetic polymers. It exists in two main forms:
High-density polyethylene (HDPE): Mostly linear molecules that pack closely together, used for milk jugs, bottle caps, and toys.
Low-density polyethylene (LDPE): Highly branched molecules, used for plastic bags, films, and wire insulation.
Thermoplastic and Thermosetting Polymers
Polymers can be classified based on their response to heat:
Thermoplastic polymers: Soften upon heating and can be remolded (e.g., polyethylene).
Thermosetting polymers: Harden permanently when formed and do not soften upon reheating; they decompose instead.
Addition Polymers
Mechanism and Examples
Addition polymerization occurs when monomers add together without the loss of any atoms, forming polymers that contain all the atoms of the monomers. Many common plastics are addition polymers.
Polyethylene: Formed from ethylene monomers.
Polypropylene: Formed from propylene monomers.
Polystyrene: Formed from styrene monomers.
Polyvinyl chloride (PVC): Formed from vinyl chloride monomers.
Polytetrafluoroethylene (PTFE or Teflon): Formed from tetrafluoroethylene monomers.
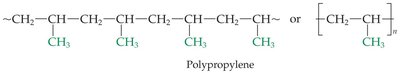
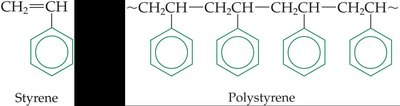
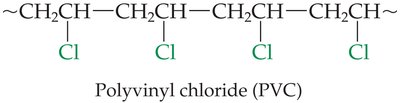
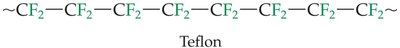

Table: Common Addition Polymers and Their Uses
Monomer | Polymer Name | Some Uses |
|---|---|---|
Ethylene (CH2=CH2) | Polyethylene | Bags, bottles, toys, electrical insulation |
Propylene (CH2=CHCH3) | Polypropylene | Carpeting, bottles, luggage |
Styrene (CH2=CH–C6H5) | Polystyrene | Insulation, cups, toys, packing materials |
Vinyl chloride (CH2=CHCl) | Polyvinyl chloride (PVC) | Food wrap, plumbing, garden hoses, floor tile |
Tetrafluoroethylene (CF2=CF2) | Polytetrafluoroethylene (Teflon) | Nonstick coatings, electrical insulation |
Conducting Polymers
Some polymers, such as polyacetylene, contain conjugated double bonds and can conduct electricity.
Molding and Processing of Plastics
Common Molding Techniques
Compression molding: Heat and pressure are applied directly to polymer powder in a mold.
Injection molding: Plastic is melted and forced into cold molds to set.
Extrusion molding: Molten polymer is extruded through a die and cut or coiled.
Blow molding: A bubble of molten polymer is blown inside a mold to form hollow objects like bottles.
Elastomers and Rubber
Natural Rubber and Vulcanization
Elastomers are polymers that stretch under force and return to their original shape. Natural rubber is an elastomer made from isoprene units. Vulcanization, discovered by Charles Goodyear, involves cross-linking rubber with sulfur to make it harder and more durable.
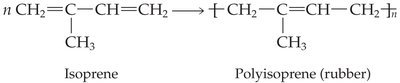
Synthetic Rubber
Synthetic rubbers are designed to mimic or improve upon natural rubber. Examples include:
Polybutadiene
Neoprene: Contains chlorine, making it more resistant to oils and solvents.
Styrene–butadiene rubber (SBR): A copolymer of styrene and butadiene, used in tires and other products.
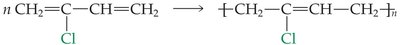
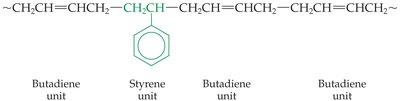
Elastomers in Paints
Elastomeric binders in paints increase resistance to cracking. Latex paints use water as a solvent, making them more environmentally friendly than oil-based paints.

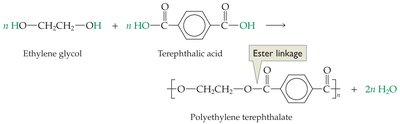
Condensation Polymers
Mechanism and Examples
Condensation polymerization involves the joining of monomers with the release of small molecules such as water, alcohols, ammonia, or HCl. Common condensation polymers include:
Nylon (polyamide): Used mainly as fibers.
Polyesters: Made from molecules containing alcohol and carboxylic acid groups; the linkage is an ester.
Phenol–formaldehyde resin (Bakelite): The first synthetic polymer.
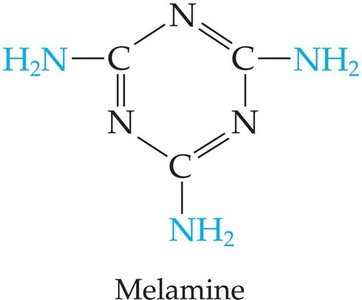
Urea–formaldehyde and melamine–formaldehyde resins: Used in adhesives and plastics.
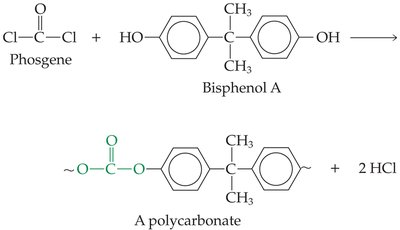
Polycarbonates: Tough, clear polymers used in safety equipment.
Polyurethanes: Used in foam rubber, skate wheels, and furniture finishes.
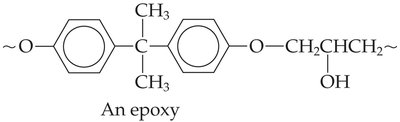
Epoxy resins: Durable adhesives and coatings, often sold as two-part liquids.
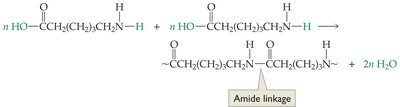
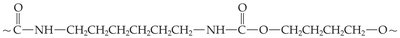
Composite Materials
Composite materials consist of high-strength fibers (such as fiberglass, graphite, or ceramics) embedded in a polymeric matrix. They are used in boat hulls, tennis rackets, automobile panels, and fishing rods.

Silicones
Structure and Properties
Silicones are polymers with silicon-oxygen backbones instead of carbon. They can be oils, gels, or elastomers, depending on their structure and degree of cross-linking.

Properties of Polymers
Molecular Weight and Structure
Polymers have high molecular weights, resulting in strong intermolecular forces and the ability to form strong fibers and viscous solutions. The arrangement of polymer chains affects their properties:
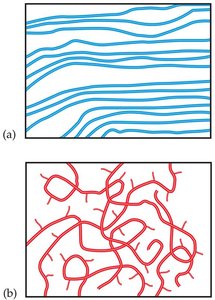
Crystalline polymers: Chains line up in neat rows, making the material rigid and strong.
Amorphous polymers: Chains are randomly tangled, making the material soft and rubbery.
Glass Transition Temperature (Tg)
The glass transition temperature is the point above which a polymer is tough and rubbery, and below which it is hard, brittle, and glass-like.
Polymer Fibers
Many synthetic polymers can be spun into fibers with properties superior to natural fibers. Most fibers and fabrics in the United States are synthetic.

Plastics and the Environment
Disposal Methods
Landfills: Plastics make up a significant portion of solid waste by mass and volume. Landfill space is limited.
Incineration: Plastics can be burned for energy, but may release toxic gases (e.g., HCl from PVC).
Degradable Plastics: Biodegradable and photodegradable plastics are being developed, especially for packaging.
Recycling: Plastics can be separated, chopped, melted, and remolded or spun into fibers.

Fire Hazards
Some plastics are flammable and may release toxic gases when burned. Regulations require flame-retardant materials for certain uses, such as children's sleepwear.
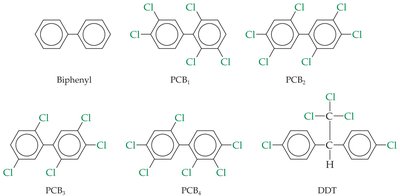
Plasticizers and Pollution
Plasticizers are added to make plastics more flexible. Early plasticizers, such as polychlorinated biphenyls (PCBs), were toxic. Modern plasticizers, such as phthalate esters, are less toxic but still a concern for environmental pollution.