 Back
BackStoichiometry and Quantities in Chemical Reactions
Study Guide - Smart Notes

Ch. 8: Quantities in Chemical Reactions
Stoichiometry
Stoichiometry is the study of the numerical relationships between the quantities of reactants and products in a balanced chemical equation. It allows chemists to predict the amount of product that can be formed from a given amount of reactant, or vice versa.
Balanced chemical equations provide the mole ratios needed for calculations.
Stoichiometry is essential for laboratory work, industrial processes, and understanding chemical reactions quantitatively.
Key concept: The coefficients in a balanced equation represent the relative number of moles of each substance involved in the reaction.
Mass-to-Mass Conversions
Mass-to-mass conversions involve calculating the mass of one substance in a reaction from the known mass of another substance. This process uses molar masses and stoichiometric relationships from the balanced equation.
Step 1: Convert the mass of the given substance (A) to moles using its molar mass.
Step 2: Use the mole ratio from the balanced equation to convert moles of A to moles of B.
Step 3: Convert moles of B to mass using the molar mass of B.
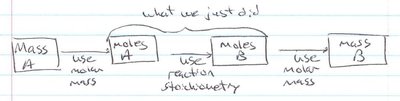
Example: If you start with 10.0 g of A and want to find the mass of B produced, follow the steps above using the appropriate molar masses and stoichiometric coefficients.
Limiting Reactant (Limiting Reagent)
The limiting reactant is the reactant that is completely consumed first in a chemical reaction, thus determining the maximum amount of product that can be formed. The other reactant(s) are in excess.
To identify the limiting reactant, calculate the amount of product that can be formed from each reactant. The reactant that produces the least amount of product is the limiting reactant.
Theoretical Yield, Actual Yield, and Percent Yield
Theoretical yield is the maximum amount of product that can be produced from the limiting reactant, as calculated from stoichiometry. Actual yield is the amount of product actually obtained from the reaction. Percent yield compares the actual yield to the theoretical yield and is calculated as follows:
Theoretical yield: Calculated from the limiting reactant using stoichiometry.
Actual yield: Measured experimentally.
Percent yield:

Example: For the reaction , if you have 53.2 g Na and 65.8 g Cl2, and your actual yield is 86.4 g, you can:
Find the limiting reagent by calculating the moles of NaCl that can be formed from each reactant.
Calculate the theoretical yield in grams of NaCl.
Determine the percent yield using the formula above.
Enthalpy of a Reaction ()
The enthalpy change of a reaction () is the heat absorbed or released during a chemical reaction at constant pressure. The sign of $\Delta H_{\text{RXN}}$ indicates the direction of heat flow:
Exothermic reaction: (heat is released)
Endothermic reaction: (heat is absorbed)
can be used as a conversion factor in stoichiometric calculations to determine the total heat absorbed or released based on the amount of reactant consumed:
Use the balanced equation and to relate moles of reactant to energy change.
Example calculation: If kJ for the formation of 1 mol of H2O, then forming 2 mol of H2O releases kJ of heat.
Key formula:
